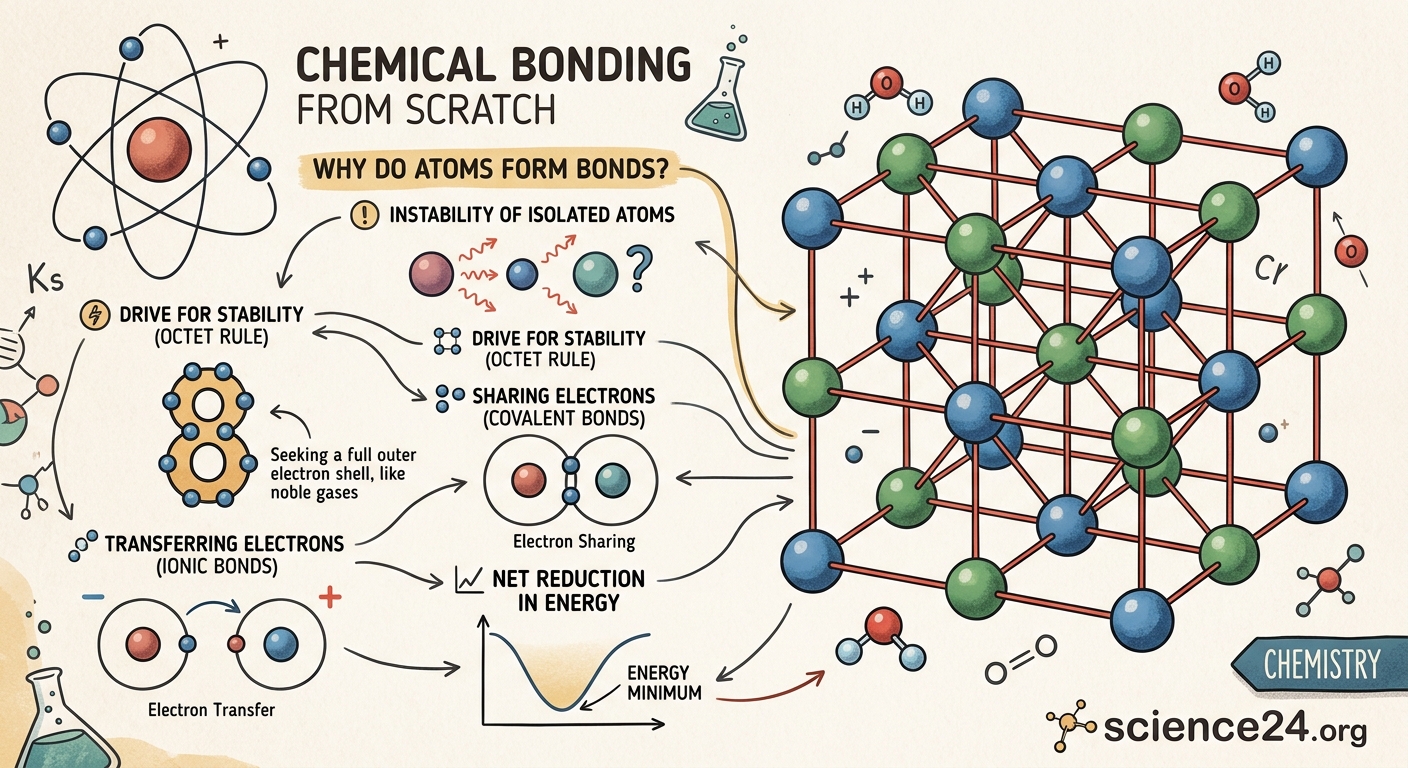
Atoms are the building blocks of everything around you, but they rarely exist alone in nature. Instead, they join together to form molecules, compounds, and all the materials you interact with daily. This tendency to connect isn’t random. It’s driven by fundamental principles of energy and stability that govern the atomic world.
Atoms form bonds to achieve lower energy states and greater stability. By sharing, transferring, or pooling electrons, atoms reach electron configurations similar to noble gases. This process releases energy, making bonded atoms more stable than isolated ones. Chemical bonding explains why molecules exist and how matter assembles into everything we see.
Energy Drives Everything Atoms Do
At the heart of chemical bonding lies a simple principle that energy likes to minimize. Atoms naturally move toward configurations that lower their total energy. Think of a ball rolling downhill. It doesn’t need a push to descend because gravity pulls it toward the lowest point. Atoms behave similarly with their electron arrangements.
Isolated atoms carry higher potential energy than bonded atoms. When two hydrogen atoms approach each other, their electrons begin interacting with both nuclei. This arrangement creates a lower energy state than either atom had alone. The difference in energy gets released, usually as heat, and the atoms settle into a stable bond.
This energy reduction isn’t just theoretical. You can measure it in every chemical reaction. When bonds form, energy releases. When bonds break, energy must be supplied. The drive toward lower energy explains why atoms spontaneously form bonds under the right conditions.
Electron Configurations and the Octet Rule
Atoms aren’t all equally eager to bond. Their willingness depends on their electron configuration, particularly in the outermost shell called the valence shell. Noble gases like helium, neon, and argon exist comfortably as single atoms because their valence shells are already full. They’ve achieved the lowest energy state possible for their electron count.
Other atoms lack this completeness. Oxygen has six valence electrons but needs eight for a full shell. Sodium has one electron beyond a stable inner configuration. These atoms exist in higher energy states and actively seek ways to achieve noble gas configurations.
The octet rule describes this tendency. Most atoms become stable when they have eight electrons in their valence shell, matching the configuration of noble gases. Hydrogen and helium are exceptions, needing only two electrons to fill their first shell. This rule guides how atoms bond and in what ratios.
“Chemical bonds form not because atoms ‘want’ to achieve full shells, but because the resulting electron distribution creates a lower energy state for the entire system. The octet rule is a useful shorthand for predicting this outcome.”
Three Primary Ways Atoms Achieve Stability
Atoms can reach stable electron configurations through three main bonding strategies. Each method involves electrons but operates differently:
-
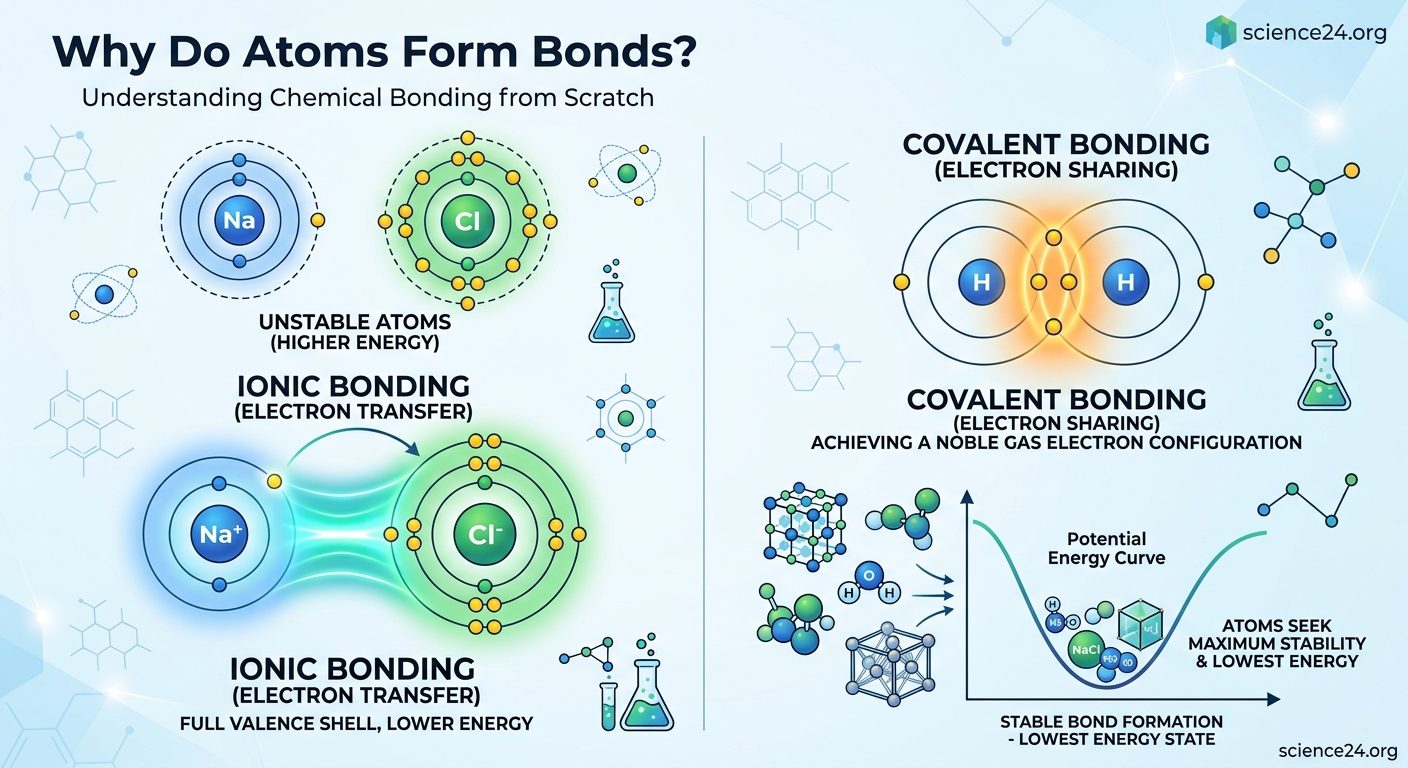
Ionic bonding occurs when one atom completely transfers electrons to another. Sodium donates its single valence electron to chlorine, which needs one electron to complete its octet. This transfer creates oppositely charged ions that attract each other electrostatically. The resulting compound, sodium chloride, is table salt.
-
Covalent bonding involves atoms sharing electrons rather than transferring them. Two hydrogen atoms each contribute one electron to form a shared pair. Both atoms effectively have two electrons, filling their shells. Water, carbon dioxide, and most organic molecules use covalent bonds.
-
Metallic bonding happens when many atoms pool their valence electrons into a shared “sea” that moves freely among positive metal ions. This arrangement explains why metals conduct electricity and can be shaped without breaking. The electrons aren’t tied to specific atoms but belong to the entire structure.
Electronegativity Determines Bond Character
Not all atoms pull on shared electrons with equal strength. Electronegativity measures an atom’s ability to attract electrons in a bond. Fluorine has the highest electronegativity, while francium has the lowest. This property determines what type of bond forms between two atoms.
When atoms with very different electronegativities meet, ionic bonds typically form. The more electronegative atom essentially strips electrons from the less electronegative one. Sodium (low electronegativity) and chlorine (high electronegativity) create a classic ionic bond.
When atoms have similar electronegativities, they share electrons more equally in covalent bonds. Two oxygen atoms have identical electronegativity, so they share electrons perfectly. Carbon and hydrogen have similar values, which is why organic molecules feature covalent bonds throughout.
Bonds exist on a spectrum. Pure ionic and pure covalent bonds are extremes. Most bonds have some ionic character and some covalent character. Water molecules demonstrate this. Oxygen pulls harder on shared electrons than hydrogen does, creating partial charges even though the bond is primarily covalent.
The Role of Nuclear Charge and Distance
Atomic nuclei play a critical role in bonding. Each proton in the nucleus attracts electrons through electromagnetic force. More protons mean stronger attraction. But electrons also repel each other, and inner shell electrons shield outer electrons from the full nuclear charge.
Distance matters enormously. Electromagnetic force decreases with the square of distance. An electron close to a nucleus experiences much stronger attraction than one farther away. This explains why valence electrons in large atoms bond differently than those in small atoms.
When two atoms approach each other, attractive and repulsive forces compete. Nuclei attract electrons from both atoms. Electrons repel other electrons. Nuclei repel each other. At a specific distance, these forces balance to create the minimum energy state. That distance becomes the bond length.
If atoms get pushed closer than the bond length, repulsion dominates and energy increases. If they’re pulled apart, attraction weakens and energy also increases. The bond represents an energy minimum, a stable equilibrium point.
Common Misconceptions About Chemical Bonding
| Misconception | Reality |
|---|---|
| Atoms “want” to achieve full shells | Atoms don’t have desires; they simply move toward lower energy states |
| Ionic bonds involve no electron sharing | Even ionic bonds have some electron cloud overlap and partial covalent character |
| Covalent bonds always share electrons equally | Only bonds between identical atoms share equally; most have polar character |
| Noble gases never form bonds | Larger noble gases can form compounds under specific conditions |
| Bonds are static structures | Electrons constantly move; bonds represent average positions and probabilities |
These misunderstandings often stem from simplified teaching models. The reality involves quantum mechanics and probability distributions. Electrons don’t orbit like planets. They exist in clouds of probability. Bonds represent regions where electron probability overlaps between atoms.
Energy Changes During Bond Formation
Let’s walk through what happens energetically when a bond forms:
-
Two isolated atoms have a baseline energy determined by their electron configurations and nuclear charges.
-
As atoms approach, attractive forces begin pulling electrons toward both nuclei, lowering the system’s potential energy.
-
At the optimal distance, attractive and repulsive forces balance, creating the lowest possible energy state for that pair of atoms.
-
The energy difference between isolated atoms and bonded atoms gets released, typically as heat or light, making the surroundings warmer.
-
To break the bond later requires supplying exactly that much energy back to the system.
This energy change is measurable and predictable. Bond energies tell chemists how much energy different bonds store. Stronger bonds require more energy to break. The carbon-carbon triple bond in acetylene stores more energy than the single bond in ethane.
Practical Applications of Bonding Principles
Understanding why atoms form bonds helps you predict chemical behavior. You can anticipate which elements will react together. Sodium and chlorine react vigorously because their bonding releases substantial energy. Noble gases don’t react under normal conditions because they’re already at low energy states.
Bond types determine material properties. Ionic compounds typically form crystalline solids with high melting points. The strong electrostatic attraction between ions requires lots of energy to overcome. Covalent compounds can be gases, liquids, or solids depending on molecular size and shape. Metals conduct electricity because their bonded electrons move freely.
You can predict molecular shapes using bonding principles. Atoms arrange themselves to minimize electron repulsion while maintaining bonds. Water bends because oxygen’s two lone pairs of electrons repel the bonding pairs. Carbon dioxide stays linear because that geometry minimizes repulsion around the carbon atom.
Chemical reactions involve breaking old bonds and forming new ones. Reactions release energy when the new bonds are stronger than the old ones. They require energy input when the opposite is true. This principle governs everything from combustion engines to cellular respiration.
Quantum Mechanics Provides the Complete Picture
Classical physics can’t fully explain chemical bonding. Electrons don’t follow predictable paths like planets. They exist in probability clouds called orbitals. When atoms bond, their orbitals overlap and combine into molecular orbitals.
Molecular orbital theory describes how atomic orbitals merge. Some combinations create bonding orbitals with electron density between nuclei, lowering energy and creating attraction. Other combinations create antibonding orbitals that increase energy. Electrons preferentially occupy bonding orbitals, stabilizing the molecule.
The mathematics gets complex, but the concept remains accessible. Electrons behave like waves. When waves overlap constructively, they reinforce each other. When they overlap destructively, they cancel out. Bonding orbitals result from constructive interference. Antibonding orbitals come from destructive interference.
This quantum mechanical view explains phenomena that simple models miss. It predicts magnetic properties, spectroscopic behavior, and reaction mechanisms. For most everyday chemistry, simpler models work fine. But the quantum picture provides the fundamental truth about why atoms form bonds.
Comparing Different Bond Types
Different bonding strategies create different material properties:
- Ionic compounds dissolve in water because polar water molecules surround and separate ions
- Covalent network solids like diamond are extremely hard because breaking the material requires breaking many strong covalent bonds
- Molecular covalent compounds often have low melting points because only weak intermolecular forces hold separate molecules together
- Metals can be drawn into wires because metallic bonds allow atoms to slide past each other without breaking the overall bonding
These differences matter practically. You wouldn’t make electrical wires from ionic compounds because they don’t conduct electricity as solids. You wouldn’t make jewelry from sodium metal because it reacts violently with water and air. Material selection depends on understanding bonding types.
Engineers and chemists use bonding knowledge to design new materials. They combine elements strategically to achieve desired properties. Semiconductors mix elements to create specific electronic behaviors. Polymers link monomers through carefully chosen covalent bonds. Alloys blend metals to balance strength, flexibility, and corrosion resistance.
Why This Matters for Your Chemistry Studies
Grasping why atoms form bonds gives you a foundation for understanding all of chemistry. Reaction mechanisms make sense when you see them as bond breaking and forming. Thermodynamics becomes clearer when you connect heat flow to bond energies. Organic chemistry’s countless reactions follow patterns based on bonding principles.
Your exam questions often test whether you understand the reasoning behind bonding, not just memorized facts. Can you predict which compound has a higher melting point? Can you explain why certain reactions release heat? Can you draw electron dot structures that show how atoms achieve stable configurations?
Practice applying these principles to unfamiliar situations. Don’t just memorize that water is polar. Understand that oxygen’s higher electronegativity creates that polarity. Don’t just remember that sodium and chlorine form ionic bonds. Recognize that their large electronegativity difference drives electron transfer.
The concepts connect to other science topics too. Energy minimization appears throughout physics, from gravitational systems to electromagnetic fields. Electron behavior relates to quantum mechanics and wave theory. The same mathematical tools that describe atomic orbitals also describe how electromagnetic induction powers your everyday devices.
Bringing Atoms Together in Your Understanding
Chemical bonding isn’t mysterious once you see the energy perspective. Atoms form bonds because doing so lowers their energy and increases their stability. Whether they transfer electrons, share them, or pool them into a metallic sea, the result is the same. The bonded state stores less energy than isolated atoms.
This principle drives the chemistry of life, the materials in your phone, the fuel in vehicles, and the air you breathe. Every molecule exists because its bonds create a more stable arrangement than separate atoms. Understanding this fundamental truth transforms chemistry from memorization into logical prediction. You’re not just learning facts. You’re learning to think like matter itself, always seeking the path to lower energy and greater stability.





