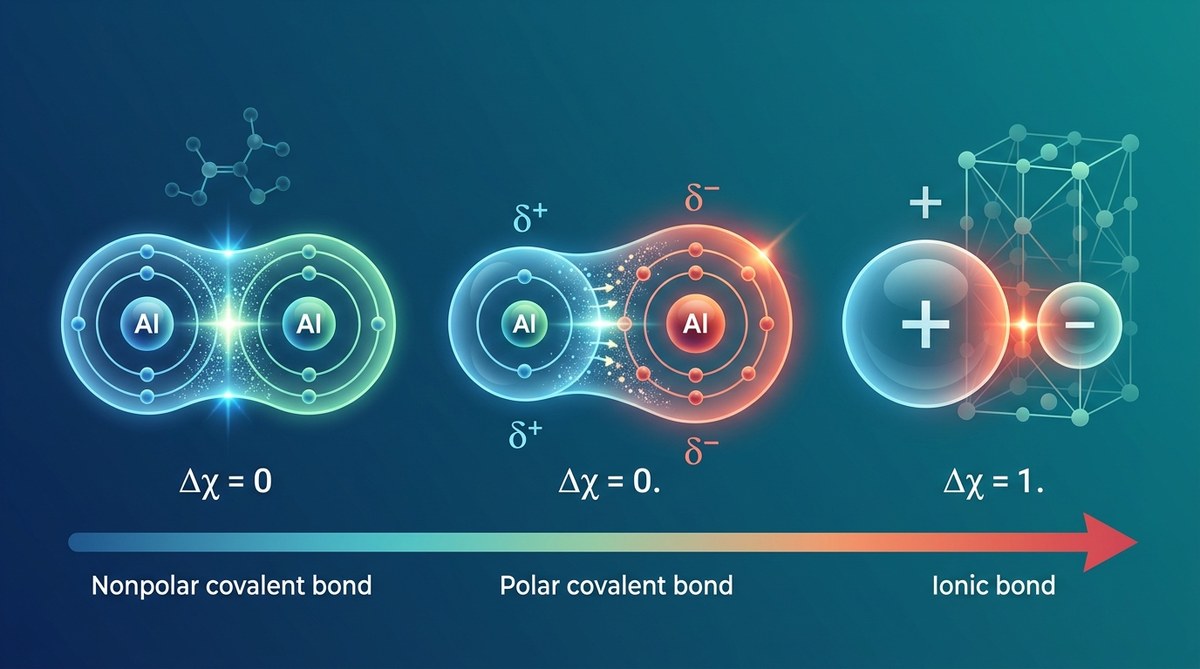
When two atoms meet, they don’t just randomly stick together. The type of bond they form depends almost entirely on one property: electronegativity. This single value tells you whether electrons will be shared equally, shared unequally, or transferred completely from one atom to another. Understanding this relationship transforms chemistry from memorization into prediction.
Electronegativity differences between atoms determine bond type. Small differences (less than 0.5) create nonpolar covalent bonds with equal electron sharing. Medium differences (0.5 to 1.7) produce polar covalent bonds with unequal sharing. Large differences (greater than 1.7) result in ionic bonds where electrons transfer completely. This pattern predicts molecular behavior and chemical properties.
What Electronegativity Actually Measures
Electronegativity measures an atom’s ability to attract electrons in a chemical bond. Linus Pauling developed the most widely used scale in the 1930s, assigning fluorine a value of 4.0 as the most electronegative element. Cesium and francium sit at the bottom with values around 0.7.
The scale isn’t arbitrary. It reflects nuclear charge, atomic radius, and electron shielding. Smaller atoms with more protons pull electrons more strongly. Fluorine, oxygen, and nitrogen dominate the high end. Metals like sodium and potassium cluster at the low end.
This numerical difference between two bonded atoms predicts everything about their relationship. The gap tells you who controls the electrons and how those electrons behave.
The Three Bond Types Explained
Chemical bonds fall into three categories based on electron behavior. Each type creates distinct properties in the resulting compound.
Nonpolar covalent bonds form when electronegativity differences stay below 0.5. Electrons spend equal time around both atoms. The H₂ molecule provides a perfect example. Both hydrogen atoms have identical electronegativity values of 2.1, creating a difference of exactly zero. The electron pair sits symmetrically between them.
Polar covalent bonds emerge when differences range from 0.5 to 1.7. Electrons still get shared, but not equally. The more electronegative atom pulls harder, creating partial charges. Water demonstrates this beautifully. Oxygen (3.5) bonds with hydrogen (2.1), producing a difference of 1.4. The oxygen end becomes slightly negative while hydrogen ends become slightly positive.
Ionic bonds occur when differences exceed 1.7. The electronegative atom strips electrons away completely. Sodium chloride shows this extreme. Chlorine (3.0) overwhelms sodium (0.9) with a difference of 2.1. Sodium loses its electron entirely, becoming Na⁺. Chlorine gains it, becoming Cl⁻. The resulting ions attract through electrostatic forces.
These cutoffs aren’t rigid laws. They’re useful guidelines that work for most situations. Some chemists use slightly different thresholds, but the pattern holds regardless.
How to Predict Bond Type Using Electronegativity Values
Follow these steps to determine what type of bond will form between any two atoms:
- Find both electronegativity values using a periodic table or reference chart.
- Subtract the smaller value from the larger value to get the absolute difference.
- Compare your result to the threshold ranges: below 0.5 for nonpolar, 0.5 to 1.7 for polar, above 1.7 for ionic.
- Identify which atom is more electronegative to determine the direction of electron pull.
- Predict molecular properties based on the bond type you identified.
Let’s apply this method to carbon dioxide. Carbon has an electronegativity of 2.5. Oxygen sits at 3.5. The difference equals 1.0, placing it firmly in polar covalent territory. Each C=O bond is polar with oxygen pulling electrons harder than carbon.
Here’s another example with calcium fluoride. Calcium measures 1.0 on the scale. Fluorine tops out at 4.0. The difference of 3.0 screams ionic bond. Calcium surrenders two electrons to become Ca²⁺. Each fluorine atom accepts one electron to become F⁻.
Why the Difference Matters More Than Individual Values
You might think atoms with high electronegativity always form certain bond types. That’s not how it works. The relationship between two atoms determines everything, not their absolute values.
Consider chlorine gas (Cl₂). Chlorine is highly electronegative at 3.0. But when two chlorine atoms bond, the difference is zero. They form a perfectly nonpolar covalent bond despite each atom’s strong electron-pulling ability.
Now look at hydrogen chloride. Hydrogen sits at 2.1 and chlorine at 3.0. The difference of 0.9 creates a polar covalent bond. Same chlorine atom, completely different bond type.
This principle explains why metals and nonmetals typically form ionic bonds. The electronegativity gap between them usually exceeds 1.7. Nonmetals bonding with other nonmetals generally stay in the covalent range because their values are closer together.
Common Patterns Across the Periodic Table
Electronegativity follows predictable trends that make bond prediction easier. Values increase from left to right across periods. They decrease from top to bottom down groups.
These patterns create zones on the periodic table. The upper right corner (excluding noble gases) contains the most electronegative elements. The lower left corner holds the least electronegative elements. Bonds between atoms from opposite corners will almost always be ionic.
Elements close together on the table tend to form covalent bonds. Elements far apart tend to form ionic bonds. This spatial relationship on the periodic table directly maps to bond type, making the beginner’s guide to reading the periodic table like a pro incredibly useful for predictions.
Real Examples That Clarify the Concepts
Let’s examine several molecules to see these principles in action:
- Methane (CH₄): Carbon (2.5) and hydrogen (2.1) differ by only 0.4. The bonds are essentially nonpolar covalent. Methane behaves as a nonpolar molecule.
- Ammonia (NH₃): Nitrogen (3.0) and hydrogen (2.1) differ by 0.9. These polar covalent bonds make ammonia a polar molecule that dissolves readily in water.
- Magnesium oxide (MgO): Magnesium (1.2) and oxygen (3.5) differ by 2.3. This massive gap creates an ionic compound with a crystal lattice structure and high melting point.
- Carbon monoxide (CO): Carbon (2.5) and oxygen (3.5) differ by 1.0. The polar covalent bond gives this molecule a slight dipole moment.
Each difference produces distinct physical and chemical properties. Nonpolar molecules don’t dissolve in water. Polar molecules do. Ionic compounds conduct electricity when melted or dissolved. These behaviors stem directly from bond type.
The Gray Area Between Bond Types
Chemistry rarely provides absolute boundaries. The transition from polar covalent to ionic happens gradually, not suddenly at 1.7.
A compound with a difference of 1.6 shows significant ionic character even though we classify it as polar covalent. A compound at 1.8 might still exhibit some covalent character. The cutoff serves as a practical tool, not a fundamental law of nature.
Some chemists calculate percent ionic character to quantify this gray area. A completely nonpolar bond has 0% ionic character. A purely ionic bond approaches 100% ionic character. Most bonds fall somewhere between these extremes.
This spectrum explains why some “ionic” compounds have lower melting points than expected or why some “covalent” compounds conduct electricity slightly when dissolved. Why do atoms form bonds? Understanding chemical bonding from scratch helps clarify these edge cases.
How Bond Type Affects Molecular Properties
The electronegativity difference doesn’t just label a bond. It determines how molecules behave in the real world.
Solubility patterns follow directly from bond polarity. Nonpolar molecules dissolve in nonpolar solvents like hexane. Polar molecules dissolve in polar solvents like water. Ionic compounds dissolve in polar solvents and dissociate into ions.
Melting and boiling points reflect bond strength and intermolecular forces. Ionic compounds generally have the highest melting points because breaking apart the crystal lattice requires enormous energy. Polar covalent compounds have moderate values. Nonpolar covalent compounds often have the lowest values.
Electrical conductivity depends on the presence of mobile charges. Ionic compounds conduct when melted or dissolved because ions move freely. Polar covalent compounds rarely conduct. Nonpolar covalent compounds never conduct.
Reactivity changes with bond polarity. Polar bonds create sites of partial charge that attract other molecules. Nonpolar bonds are less reactive. These differences explain why some reactions happen instantly while others require catalysts.
Calculating Electronegativity Differences Accurately
Students often make mistakes when calculating differences. Here’s a table showing common errors and how to avoid them:
| Mistake | Why It Happens | Correct Approach |
|---|---|---|
| Using wrong values | Consulting outdated charts | Use Pauling scale values from current periodic tables |
| Forgetting absolute value | Subtracting in wrong order | Always subtract smaller from larger |
| Rounding too early | Trying to simplify calculations | Keep full precision until final classification |
| Ignoring multiple bonds | Focusing on one pair of atoms | Check all unique bonds in polyatomic molecules |
| Misreading the scale | Confusing with other properties | Verify you’re reading electronegativity, not ionization energy |
Practice with these common pairs to build confidence:
- N and H: 3.0 minus 2.1 equals 0.9 (polar covalent)
- K and Br: 2.8 minus 0.8 equals 2.0 (ionic)
- C and C: 2.5 minus 2.5 equals 0.0 (nonpolar covalent)
- S and O: 3.5 minus 2.5 equals 1.0 (polar covalent)
Advanced Considerations for Complex Molecules
Polyatomic molecules contain multiple bonds, each with its own electronegativity difference. Carbon dioxide has two C=O bonds, both polar. But the molecule itself is nonpolar because the bond dipoles cancel due to linear geometry.
Water also has two O-H bonds, both polar. The molecule is polar because the bent geometry prevents cancellation. Molecular shape matters as much as individual bond polarity.
Resonance structures complicate the picture further. In molecules where electrons are delocalized across multiple atoms, you can’t assign a single bond type. Benzene demonstrates this with its electron cloud spread evenly around the ring.
Coordinate covalent bonds add another layer. Both electrons in the bond come from one atom, but electronegativity differences still determine polarity. Ammonia forming a bond with a proton creates this situation.
Understanding electronegativity differences is the foundation for predicting molecular behavior. Master this concept and you’ll understand why substances mix, how reactions proceed, and what properties to expect from any compound you encounter.
Practical Applications in Your Chemistry Course
Your exams will test this concept repeatedly. You’ll need to predict bond types, explain molecular polarity, and justify physical properties based on electronegativity differences.
Lab work becomes clearer when you understand bonding. Solubility experiments make sense. Conductivity tests have predictable outcomes. Reaction mechanisms follow logical patterns.
Problem sets often ask you to arrange bonds by increasing polarity. Calculate the difference for each pair, then rank them. Questions about which compounds dissolve in which solvents trace back to bond type. Ionic and polar compounds dissolve in water. Nonpolar compounds don’t.
Multiple choice questions might give you four compounds and ask which has the most ionic character. Calculate all four differences and choose the largest. Or they might ask which bond is most polar. Same process, different wording.
Free response questions require explanations. Don’t just state the difference. Explain what it means for electron distribution, partial charges, and molecular properties. Connect the number to observable behavior.
Study Strategies That Actually Work
Memorizing electronegativity values for common elements saves time on exams. Focus on these: H (2.1), C (2.5), N (3.0), O (3.5), F (4.0), Na (0.9), Cl (3.0), and Ca (1.0). These cover most introductory chemistry problems.
Create flashcards with element pairs on one side and bond type on the other. Include the calculation and reasoning. Shuffle them regularly to prevent pattern memorization.
Practice predicting properties from structure. Given a molecular formula, determine all bond types, predict polarity, estimate solubility, and justify melting point trends. This integrated approach mirrors exam questions.
Work through problems without a calculator first. Estimating differences builds intuition. You’ll recognize patterns faster and catch calculation errors more easily.
Draw Lewis structures before analyzing bonds. Visual representations help you identify all unique bonds and avoid missing pairs in complex molecules.
Making Sense of Periodic Trends
Electronegativity increases across periods because effective nuclear charge increases. More protons pull harder on valence electrons. Atomic radius decreases, bringing the nucleus closer to bonding electrons.
Electronegativity decreases down groups because atomic radius increases. Valence electrons sit farther from the nucleus. Inner electrons shield the nuclear charge more effectively.
These trends explain why fluorine is the most electronegative element. It sits in the upper right corner with a small radius and high nuclear charge. Francium occupies the opposite corner with a large radius and heavy shielding.
Noble gases don’t have assigned values because they rarely form bonds. When they do bond (like in xenon compounds), their electronegativity falls between that of halogens and other nonmetals.
Transition metals show less dramatic trends because d-orbital electrons complicate the picture. Their values cluster in the middle range, generally forming polar covalent or metallic bonds rather than ionic ones.
Connecting Electronegativity to Molecular Geometry
Bond polarity influences molecular shape. Polar bonds create regions of partial charge that repel other electron-dense areas. This affects bond angles and overall geometry.
VSEPR theory predicts shapes based on electron repulsion. But electronegativity differences tell you where electron density concentrates. In water, electrons spend more time near oxygen, making that region more negative and increasing repulsion between lone pairs.
Dipole moments arise from polar bonds arranged asymmetrically. The vector sum of individual bond dipoles determines the overall molecular dipole. Carbon dioxide has polar bonds but no molecular dipole. Water has both.
Understanding these connections helps you predict properties like boiling point. Polar molecules have higher boiling points than nonpolar molecules of similar mass because dipole-dipole interactions add to London dispersion forces.
Why This Concept Matters Beyond Your Exam
Electronegativity differences control chemistry everywhere. Biochemical reactions depend on polar bonds in amino acids. Drug design requires understanding how molecules interact through polar and nonpolar regions.
Materials science uses bond type to engineer properties. Ionic compounds make strong ceramics. Covalent networks create hard materials like diamond. Polar polymers absorb water while nonpolar ones repel it.
Environmental chemistry traces pollutant behavior through bonding. Nonpolar compounds like oils don’t dissolve in water, creating slicks. Polar compounds dissolve and disperse differently.
Your understanding of how electronegativity affects bond type becomes a lens for viewing molecular behavior. Every substance you encounter follows these rules. The salt dissolving in your water. The plastic that repels it. The proteins folding in your cells. All determined by electronegativity differences.
Building Your Chemical Intuition
Chemistry stops feeling like memorization once you grasp electronegativity. You can predict outcomes instead of looking them up. You understand why reactions happen instead of just accepting that they do.
Start applying this knowledge immediately. Look at molecular formulas and predict bond types. Check your work with a periodic table. Guess whether compounds will dissolve in water based on polarity. Verify your predictions with solubility tables.
This skill compounds over time. Each correct prediction strengthens your intuition. Soon you’ll recognize patterns instantly. Ionic bonding between metals and nonmetals becomes obvious. Polar covalent bonding in common molecules feels natural.
Your chemistry course builds on this foundation constantly. Reaction mechanisms, acid-base behavior, and organic chemistry all reference electronegativity and bond polarity. Master it now and everything else becomes easier. The numbers tell you exactly how atoms will interact, transforming chemistry from mystery into logic.