Organic chemistry can feel overwhelming when you first open the textbook. Hundreds of molecules, reactions, and structures compete for space in your brain. But here’s the good news: most of organic chemistry boils down to recognizing a few dozen functional groups and understanding how they behave. Once you master these building blocks, the rest starts to click into place.
Functional groups are specific atom arrangements that determine how organic molecules react. Learning to identify alcohols, carbonyls, amines, and other groups helps you predict chemical behavior, ace exams, and understand biological processes. This guide breaks down the most important functional groups with memory aids, practical examples, and study strategies that actually work for students.
What Makes Functional Groups So Important
A functional group is a specific cluster of atoms within a molecule that behaves predictably across different compounds. Think of them like LEGO pieces. The same red brick works the same way whether you’re building a house or a spaceship. Similarly, an alcohol group behaves like an alcohol whether it’s attached to a small methane-based molecule or a giant steroid structure.
This predictability is what makes organic chemistry learnable. You don’t need to memorize every single molecule. You just need to recognize the functional groups and understand their characteristic reactions.
When you see an OH group bonded to a carbon, you know you’re dealing with an alcohol. That alcohol will participate in certain reactions, have certain properties, and show up in certain biological contexts. The rest of the molecule matters, but the functional group drives the chemistry.
Hydrocarbons Form the Foundation
Before we get into the reactive groups, let’s talk about the backbone. Hydrocarbons contain only carbon and hydrogen. They’re the simplest organic compounds, and they serve as the framework for everything else.
Alkanes contain only single bonds between carbons. Methane, ethane, and propane are all alkanes. They’re relatively unreactive, which is why propane makes good fuel. It burns cleanly but doesn’t randomly react with other substances.
Alkenes have at least one carbon-carbon double bond. That double bond is a reactive site. Ethene (ethylene) is the simplest alkene, and it’s used to make plastics.
Alkynes contain at least one carbon-carbon triple bond. Acetylene is the most famous alkyne. It burns hot enough to cut through metal in welding torches.
Aromatic compounds feature benzene rings, those hexagonal structures with alternating double bonds. Benzene itself is toxic, but aromatic rings show up everywhere in biology and pharmaceuticals.
Understanding these basic structures helps because functional groups attach to these hydrocarbon frameworks. When you see a complex molecule, strip away the functional groups mentally, and you’ll find a hydrocarbon skeleton underneath.
Oxygen-Containing Groups Drive Biological Chemistry
Oxygen loves to form bonds with carbon, and the resulting functional groups are everywhere in living systems.
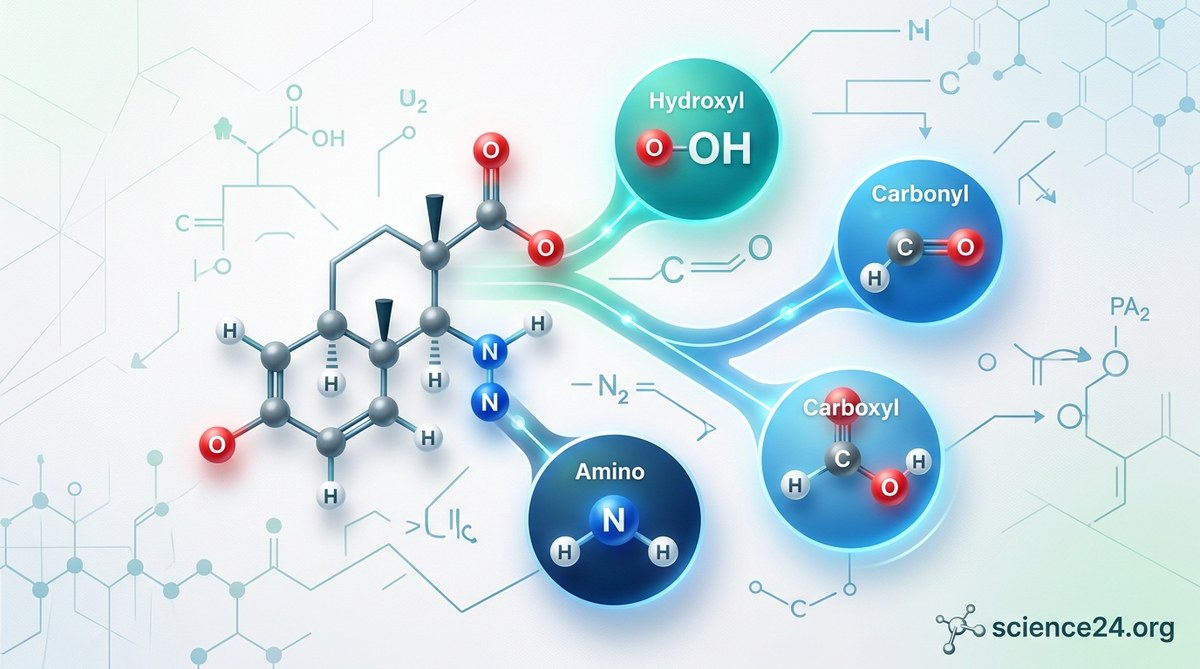
Alcohols have an OH group bonded to a carbon. Ethanol, the alcohol in beverages, is the most familiar example. Alcohols are polar, which means they mix well with water. They also participate in dehydration reactions, where water is removed to form alkenes.
Ethers contain an oxygen atom bonded to two carbon atoms (R-O-R). Diethyl ether was once used as an anesthetic. Ethers are less reactive than alcohols because they lack that hydrogen on the oxygen.
Aldehydes have a carbonyl group (C=O) with at least one hydrogen attached to the carbon. Formaldehyde is an aldehyde, though not one you want to encounter outside a lab. Aldehydes are reactive and often smell distinctive.
Ketones also have a carbonyl group, but both bonds from the carbon go to other carbons, not hydrogen. Acetone is a ketone. It’s the main ingredient in nail polish remover. Ketones are slightly less reactive than aldehydes.
Carboxylic acids combine a carbonyl with an OH group on the same carbon. Acetic acid gives vinegar its sour taste. These groups are acidic, meaning they readily donate a proton (H+) in solution.
Esters form when a carboxylic acid reacts with an alcohol. They often smell fruity. Ethyl acetate smells like pears. Esters are common in flavors and fragrances.
“Students who learn to spot carbonyl groups first find organic chemistry much easier. That C=O bond is the key to understanding aldehydes, ketones, carboxylic acids, esters, and amides. Master that one structural feature, and you’ve unlocked a huge portion of the course.” – Organic Chemistry Teaching Assistant
Nitrogen Brings Basicity and Biological Function
Nitrogen-containing functional groups are critical in biochemistry. Amino acids, proteins, and DNA all depend on nitrogen.
Amines are organic derivatives of ammonia. They have a nitrogen bonded to one, two, or three carbon groups. Amines are basic, meaning they accept protons. They often smell fishy or unpleasant. Trimethylamine gives old fish its characteristic odor.
Amides combine a carbonyl with a nitrogen. The peptide bonds that link amino acids together in proteins are amides. This functional group is incredibly important in biology. Amides are less basic than amines because the carbonyl pulls electron density away from the nitrogen.
Nitriles have a carbon triple-bonded to nitrogen (C≡N). They’re less common in introductory courses but show up in some pharmaceuticals and synthetic materials.
How to Study Functional Groups Without Losing Your Mind
Memorizing functional groups feels tedious, but there are strategies that make it manageable.
- Draw the structures repeatedly. Don’t just read them. Grab a blank sheet and draw each functional group from memory. Do this daily for a week.
- Create flashcards with the structure on one side and the name plus key properties on the other. Include an example molecule for each group.
- Use mnemonics for tricky ones. “Ketones have carbonyl groups stuck in the middle” helps you remember that ketones have carbon groups on both sides of the carbonyl.
- Group similar structures together. Study all the carbonyl-containing groups in one session. Notice what makes each one unique.
- Connect functional groups to real molecules you know. Ethanol is an alcohol. Acetone is a ketone. Aspirin contains a carboxylic acid and an ester.
The pattern recognition skill you develop here pays off throughout the entire course. Just like learning to recognize patterns helps with mental math, recognizing functional groups makes complex molecules manageable.
Common Mistakes Students Make With Functional Groups
| Mistake | Why It Happens | How to Fix It |
|---|---|---|
| Confusing aldehydes and ketones | Both have carbonyl groups | Remember: aldehydes have at least one H on the carbonyl carbon, ketones have two carbon groups |
| Missing functional groups in complex molecules | Getting distracted by the size | Circle or highlight functional groups first, then analyze the rest |
| Forgetting that some molecules have multiple groups | Focusing on just one feature | Always scan the entire structure systematically |
| Mixing up ethers and esters | Similar names | Ethers are R-O-R (simple), esters have a carbonyl next to the oxygen |
| Assuming all OH groups are alcohols | Not checking what the oxygen is bonded to | Check if the OH is on a carbonyl (carboxylic acid) or a benzene ring (phenol) |
Less Common Groups You’ll Still Encounter
Some functional groups show up less frequently but are still important for exams and advanced topics.
Thiols are the sulfur version of alcohols (R-SH). They smell terrible. Skunk spray contains thiols. But they’re also important in proteins, where cysteine residues form disulfide bonds.
Sulfides are the sulfur version of ethers (R-S-R). Garlic and onions owe their pungent flavors to sulfide-containing compounds.
Phosphates contain phosphorus bonded to oxygen atoms. They’re crucial in biochemistry. ATP, the energy currency of cells, is loaded with phosphate groups.
Acid chlorides have a carbonyl bonded to a chlorine atom. They’re extremely reactive and useful in synthesis but dangerous to handle.
Anhydrides form when two carboxylic acids lose a water molecule and bond together. Acetic anhydride is used to make aspirin.
Functional Groups in Biological Molecules
Understanding functional groups organic chemistry becomes especially important when you study biochemistry. Every major biological molecule is defined by its functional groups.
Carbohydrates are loaded with alcohol groups and often have aldehyde or ketone groups. Glucose, the sugar in your blood, is technically an aldehyde.
Lipids (fats) contain long hydrocarbon chains with ester linkages. That’s why soap, which breaks apart esters, can dissolve grease.
Proteins are chains of amino acids connected by amide bonds. The side chains on those amino acids contain various functional groups that determine protein structure and function.
Nucleic acids like DNA contain phosphate groups, ether linkages in the sugar backbone, and nitrogen-containing bases with multiple amine groups.
When you understand the functional groups, you understand why these molecules behave the way they do. You can predict how enzymes might interact with substrates, how drugs bind to receptors, and how metabolic pathways transform one molecule into another.
Similar to how understanding chemical bonding from scratch helps you grasp molecular structure, recognizing functional groups helps you understand reactivity.
Reactivity Patterns You Need to Know
Different functional groups react in characteristic ways. Here are the patterns that show up most often on exams.
- Alcohols can be oxidized to aldehydes or ketones, depending on their structure
- Aldehydes oxidize easily to carboxylic acids
- Ketones resist oxidation, making them more stable than aldehydes
- Carboxylic acids react with alcohols to form esters
- Amines act as bases and nucleophiles in many reactions
- Alkenes undergo addition reactions across the double bond
- Aromatic rings resist addition but undergo substitution reactions
These patterns repeat throughout your course. A reaction that works on one alcohol will generally work on other alcohols. A reagent that adds across one alkene double bond will add across others.
Memory Tricks That Actually Help
Students who succeed in organic chemistry develop personal systems for remembering structures. Here are techniques that work.
The carbonyl family tree: Draw a C=O at the top. Branch down to show what happens when you add different groups. One hydrogen gives you an aldehyde. Two carbons give you a ketone. An OH gives you a carboxylic acid. An OR gives you an ester. An NH2 gives you an amide.
The oxygen ladder: Alcohols are at the bottom (most reduced). Aldehydes are one step up. Carboxylic acids are at the top (most oxidized). This helps you remember oxidation states.
Suffix patterns: Alkanes end in -ane. Alkenes end in -ene. Alkynes end in -yne. Alcohols end in -ol. Aldehydes end in -al. Ketones end in -one. Carboxylic acids end in -oic acid. The systematic names encode the functional group.
Physical property connections: Alcohols mix with water because they can hydrogen bond. Hydrocarbons don’t mix with water because they can’t. Amines smell bad because they’re volatile and basic. These connections make the groups more memorable.
Study Strategies for Exam Success
Functional groups appear on every organic chemistry exam. Here’s how to prepare effectively.
Make a master sheet with all the functional groups, their structures, and key properties. Keep it to one page. Review it before every study session.
Practice identifying functional groups in complex molecules. Your textbook has hundreds of structures. Pick random ones and circle every functional group you can find.
Work practice problems that ask you to name compounds or draw structures from names. This forces you to really understand the groups, not just recognize them passively.
Form a study group and quiz each other. Take turns drawing a structure and having the others identify the functional groups and predict properties.
Connect functional groups to lab experiences if you have them. Seeing the actual chemicals, smelling them (when safe), and observing their reactions makes the concepts stick better than just reading about them.
Functional Groups and Spectroscopy
Later in your course, you’ll learn spectroscopy techniques that identify functional groups in unknown compounds. Infrared spectroscopy shows characteristic peaks for different groups. The sharp peak around 1700 cm⁻¹ indicates a carbonyl. A broad peak around 3300 cm⁻¹ suggests an alcohol or amine.
Nuclear magnetic resonance (NMR) spectroscopy reveals the chemical environment of hydrogen and carbon atoms. Different functional groups produce different signal patterns.
Understanding functional groups now makes spectroscopy much easier later. You’ll already know what groups to look for and what properties they should have.
Why This Knowledge Transfers Beyond Chemistry Class
The functional groups you learn in organic chemistry aren’t just for exams. They show up in medicine, materials science, environmental chemistry, and everyday life.
Pharmaceuticals work because their functional groups interact with specific sites in your body. Aspirin’s carboxylic acid and ester groups are essential to its function. Antibiotics often contain multiple amine and carbonyl groups that let them bind to bacterial proteins.
Polymers like nylon and polyester are named for their functional groups. Understanding ester linkages helps you understand why polyester fabrics behave the way they do.
Environmental chemistry revolves around functional groups. The carboxylic acid groups on fatty acids make soap work. The alcohol groups on ethanol make it mix with water and gasoline.
Even cooking involves functional group chemistry. The Maillard reaction that browns meat involves carbonyl groups reacting with amines. Caramelization transforms sugar molecules by rearranging their alcohol and carbonyl groups.
Building Your Functional Group Fluency
Mastery comes from repeated exposure and active practice. Don’t expect to memorize everything in one night.
Start with the most common groups: alcohols, aldehydes, ketones, carboxylic acids, esters, amines, and amides. Get those down cold before worrying about the less common ones.
Use spaced repetition. Review your flashcards daily for the first week, then every other day, then weekly. This pattern helps move information into long-term memory.
Test yourself in different ways. Sometimes identify groups from structures. Sometimes draw structures from names. Sometimes predict properties or reactions.
Connect new groups to ones you already know. When you learn about acid chlorides, connect them to carboxylic acids. When you learn about thiols, connect them to alcohols.
Turning Functional Groups Into Second Nature
The goal isn’t just to pass the next exam. The goal is to develop chemical intuition, where you can look at a molecule and immediately understand its behavior. That intuition comes from functional group recognition.
Professional chemists don’t consciously think “that’s an alcohol with an OH group.” They just see it and know. You’ll get there too with enough practice.
Start by identifying functional groups in every molecule you encounter in your textbook, lecture slides, and homework. Make it automatic. Before you even read the question, circle the functional groups.
As you move through your course, you’ll see the same groups in different contexts. Alcohols in one chapter, then in synthesis problems, then in biological molecules, then in spectroscopy. Each encounter reinforces your understanding.
The students who struggle in organic chemistry are often the ones who never fully mastered functional groups. They try to memorize individual reactions without understanding the underlying patterns. Don’t make that mistake.
Your Functional Group Mastery Starts Now
Functional groups organic chemistry doesn’t have to be the nightmare part of your course. With the right approach, it becomes the foundation that makes everything else easier.
Print out a functional group reference sheet and keep it visible while you study. Draw the structures until you can reproduce them from memory. Connect each group to real molecules and real applications. Use the memory tricks that work for your learning style.
Most importantly, start now. Don’t wait until the week before the exam. Spend 15 minutes each day reviewing functional groups, and by exam time, they’ll be second nature. You’ll walk into that test room confident, ready to identify any structure they throw at you, and prepared to predict how those molecules will behave. That confidence makes all the difference between struggling through organic chemistry and actually enjoying the elegant logic of how molecules work.