You’re staring at a chemistry problem that asks for the molarity of a solution, and the numbers seem to blur together. You know you need moles and volume, but how do they fit together? Understanding molarity is one of those fundamental skills that unlocks nearly every solution-based chemistry problem you’ll face.
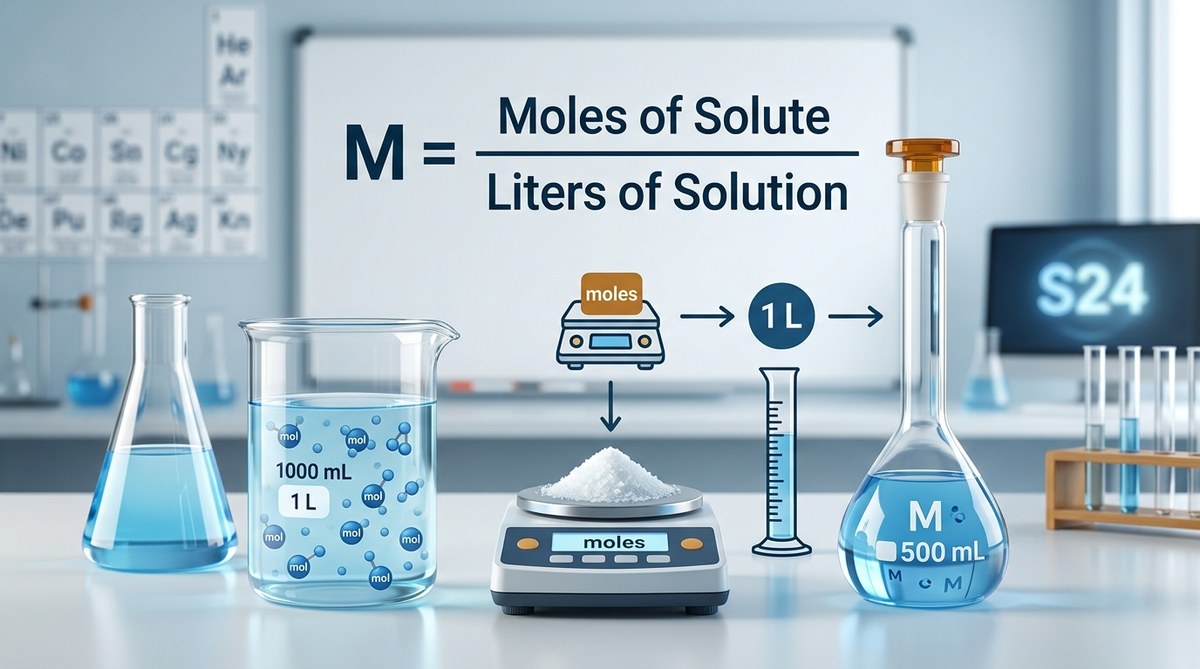
Molarity measures solution concentration as moles of solute per liter of solution. The formula M = n/V lets you calculate concentration when you know how much substance dissolves in a given volume. Mastering molarity calculations requires careful unit conversion, understanding the difference between solute mass and moles, and recognizing that volume refers to the final solution, not just the solvent added.
Defining Molarity in Simple Terms
Molarity tells you how concentrated a solution is by counting particles at the molecular level.
The official definition states that molarity (M) equals the number of moles of solute divided by the volume of solution in liters. One mole contains approximately 6.022 × 10²³ particles, known as Avogadro’s number.
Think of molarity as a ratio that answers this question: how many moles of dissolved substance exist in every liter of the final mixture?
A 1 M solution of sodium chloride means exactly one mole of NaCl dissolves in enough water to make one liter total. The water volume you add might be slightly less than one liter because the salt itself takes up space.
This distinction trips up many students. You’re measuring the final solution volume, not the solvent volume you started with.
The Core Formula and What Each Variable Means
The molarity equation looks deceptively simple:
M = n / V
Here’s what each symbol represents:
- M stands for molarity, measured in moles per liter (mol/L)
- n represents moles of solute, the substance being dissolved
- V indicates volume of the solution in liters
You’ll sometimes see molarity written with a capital M after the number, like 0.5 M or 2.0 M. The unit “molar” is interchangeable with mol/L.
Converting between grams and moles requires the molar mass of your solute. You find this by adding up the atomic masses of all atoms in the chemical formula, using values from the beginner’s guide to reading the periodic table like a pro.
The relationship looks like this:
moles = mass (g) / molar mass (g/mol)
Once you have moles, plug them into the molarity formula along with your volume in liters.
Step-by-Step Calculation Process
Here’s how to calculate molarity from start to finish:
- Identify the solute and find its molar mass from the periodic table
- Convert the mass of solute from grams to moles using the molar mass
- Convert the solution volume to liters if given in milliliters
- Divide moles by liters to get molarity
Let’s work through a real example. You dissolve 58.5 grams of sodium chloride (NaCl) in water to make 500 mL of solution.
First, calculate the molar mass of NaCl. Sodium has an atomic mass of 23.0 g/mol, and chlorine is 35.5 g/mol. Added together, NaCl has a molar mass of 58.5 g/mol.
Next, convert grams to moles:
58.5 g ÷ 58.5 g/mol = 1.0 mol
Then convert milliliters to liters:
500 mL × (1 L / 1000 mL) = 0.500 L
Finally, calculate molarity:
M = 1.0 mol / 0.500 L = 2.0 M
Your solution has a concentration of 2.0 molar.
Common Calculation Mistakes and How to Avoid Them
Students make predictable errors when working with molarity. Recognizing these patterns helps you catch mistakes before they cost points on exams.
| Mistake | Why It Happens | How to Fix It |
|---|---|---|
| Using solvent volume instead of solution volume | Confusion about what volume to measure | Always measure total solution volume after mixing |
| Forgetting to convert mL to L | Working too fast through unit conversions | Write out conversion factors explicitly |
| Using mass instead of moles | Skipping the molar mass step | Always convert grams to moles first |
| Incorrect molar mass calculation | Arithmetic errors or missing atoms | Double-check your periodic table values |
| Rounding too early | Trying to simplify calculations | Keep extra digits until the final answer |
The volume mistake causes the most problems. When you add 50 grams of sugar to 1 liter of water, your final solution volume exceeds 1 liter because the sugar molecules occupy space. Professional chemists use volumetric flasks marked at specific volumes to ensure accuracy.
Another frequent error involves compound formulas with subscripts. Calcium chloride (CaCl₂) contains one calcium atom and two chlorine atoms. Students sometimes forget to multiply chlorine’s atomic mass by two.
Similar to common mistakes students make when balancing chemical equations, molarity errors often stem from rushing through foundational steps.
Working Backward from Molarity to Find Mass or Volume
Sometimes you know the molarity and need to find how much solute to add or what volume to prepare.
Rearranging the molarity formula gives you:
n = M × V
mass = n × molar mass
Let’s say you need to prepare 250 mL of a 0.10 M glucose (C₆H₁₂O₆) solution for a biology experiment.
First, convert volume to liters:
250 mL = 0.250 L
Calculate moles needed:
n = 0.10 mol/L × 0.250 L = 0.025 mol
Find the molar mass of glucose:
(6 × 12.0) + (12 × 1.0) + (6 × 16.0) = 180.0 g/mol
Calculate mass required:
mass = 0.025 mol × 180.0 g/mol = 4.5 g
You would weigh out 4.5 grams of glucose and add water until the total volume reaches 250 mL.
When preparing solutions in the lab, always add solute to solvent, not the other way around. This prevents dangerous exothermic reactions and ensures proper mixing. Add water gradually while stirring until you reach the target volume on your volumetric flask.
Dilution Calculations Using Molarity
Diluting a concentrated solution to a lower molarity is a standard lab technique.
The dilution formula states:
M₁V₁ = M₂V₂
Where:
– M₁ is the initial molarity
– V₁ is the initial volume
– M₂ is the final molarity
– V₂ is the final volume
This equation works because the number of moles stays constant during dilution. You’re just spreading the same amount of solute through more solution.
Suppose you have 50 mL of 6.0 M hydrochloric acid and need to dilute it to 1.0 M. What final volume should you prepare?
M₁V₁ = M₂V₂
(6.0 M)(50 mL) = (1.0 M)(V₂)
V₂ = 300 mL
You would add water to your 50 mL of concentrated acid until the total volume reaches 300 mL.
Safety note: always add acid to water, never water to acid. The heat generated can cause dangerous splashing if you pour water into concentrated acid.
Practical Applications in Chemistry and Beyond
Molarity appears throughout chemistry coursework and real-world applications.
In titration experiments, you use a solution of known molarity to determine the concentration of an unknown solution. The point where the reaction completes tells you exactly how many moles reacted.
Biochemistry relies heavily on molarity for enzyme kinetics and cellular processes. Cells maintain specific molar concentrations of ions like sodium, potassium, and calcium to function properly.
Pharmaceutical companies calculate drug concentrations in molarity to ensure accurate dosing. An IV solution might contain 0.15 M sodium chloride, matching the osmotic pressure of blood.
Environmental chemists measure pollutant concentrations in molarity to assess water quality. A lake with 0.001 M dissolved copper indicates potential toxicity to aquatic life.
These applications show why precision matters. Small calculation errors can mean the difference between a successful experiment and wasted materials or, in medical settings, patient safety issues.
Other Concentration Units and When to Use Them
Molarity isn’t the only way to express concentration. Different situations call for different units.
Molality (m) measures moles of solute per kilogram of solvent. Unlike molarity, molality doesn’t change with temperature because mass stays constant while volume expands or contracts.
Mass percent shows grams of solute per 100 grams of solution. This unit appears frequently on household product labels.
Parts per million (ppm) expresses extremely dilute concentrations, common in environmental testing and trace element analysis.
Normality (N) accounts for the number of reactive units in acid-base and redox reactions. A 1 N solution of sulfuric acid differs from 1 M because each molecule can donate two protons.
Molarity remains the most common unit in academic chemistry because it directly relates to the stoichiometry of chemical reactions. When you balance equations and perform calculations, moles provide the counting unit that makes the math work.
Building Confidence Through Practice Problems
The only way to truly master molarity is by working through varied problems.
Start with straightforward calculations where you’re given mass and volume. Then progress to problems requiring multiple steps, like finding the volume needed to prepare a specific concentration.
Practice converting between different units. If a problem gives you 2500 mg of solute and asks for molarity in a 100 mL solution, you need to convert milligrams to grams and milliliters to liters.
Try working backward from answers. If a solution is 0.50 M and contains 0.25 moles, what must the volume be? This reverse thinking strengthens your understanding of the relationships between variables.
Create your own problems using household chemicals. Look up the molar mass of table salt, sugar, or baking soda. Calculate what molarity you’d get if you dissolved a tablespoon in a cup of water. (Don’t actually drink these solutions!)
Time yourself on practice problems to build speed for exams. Many students understand molarity conceptually but struggle to complete calculations within test time limits, much like those who benefit from mental math tricks that transform calculation speed.
Making Molarity Second Nature
Molarity becomes intuitive once you stop seeing it as an abstract formula and start thinking of it as a practical counting system. Every time you calculate molarity, you’re answering a simple question: how many molecular packages fit in this container?
The formula M = n/V gives you a reliable tool for any solution problem you encounter. Write it at the top of your exam paper. Tattoo it on your brain. Let it become automatic.
Pay attention to units, convert everything to moles and liters, and double-check your arithmetic. These habits prevent the vast majority of errors. When you approach molarity with confidence and careful attention to detail, those chemistry problems that once seemed impossible start to feel manageable.
Your next lab report, homework assignment, or exam will test your ability to calculate concentrations accurately. You now have the framework to handle those challenges successfully.