Light behaves in ways that seem impossible when you first encounter them. For centuries, physicists believed light traveled purely as waves, spreading through space like ripples on water. Then came a simple experiment that shattered this understanding and forced science to accept something radical: light also acts as a stream of particles. The photoelectric effect demonstrated this dual nature so clearly that it became the foundation of quantum mechanics and earned Albert Einstein his Nobel Prize.
The photoelectric effect proves light carries energy in discrete packets called photons rather than continuous waves. When light strikes certain metals, it ejects electrons only if the light frequency exceeds a specific threshold, regardless of intensity. This phenomenon cannot be explained by wave theory alone and reveals the particle nature of light, establishing wave-particle duality as a fundamental principle of quantum mechanics.
What the photoelectric effect actually is
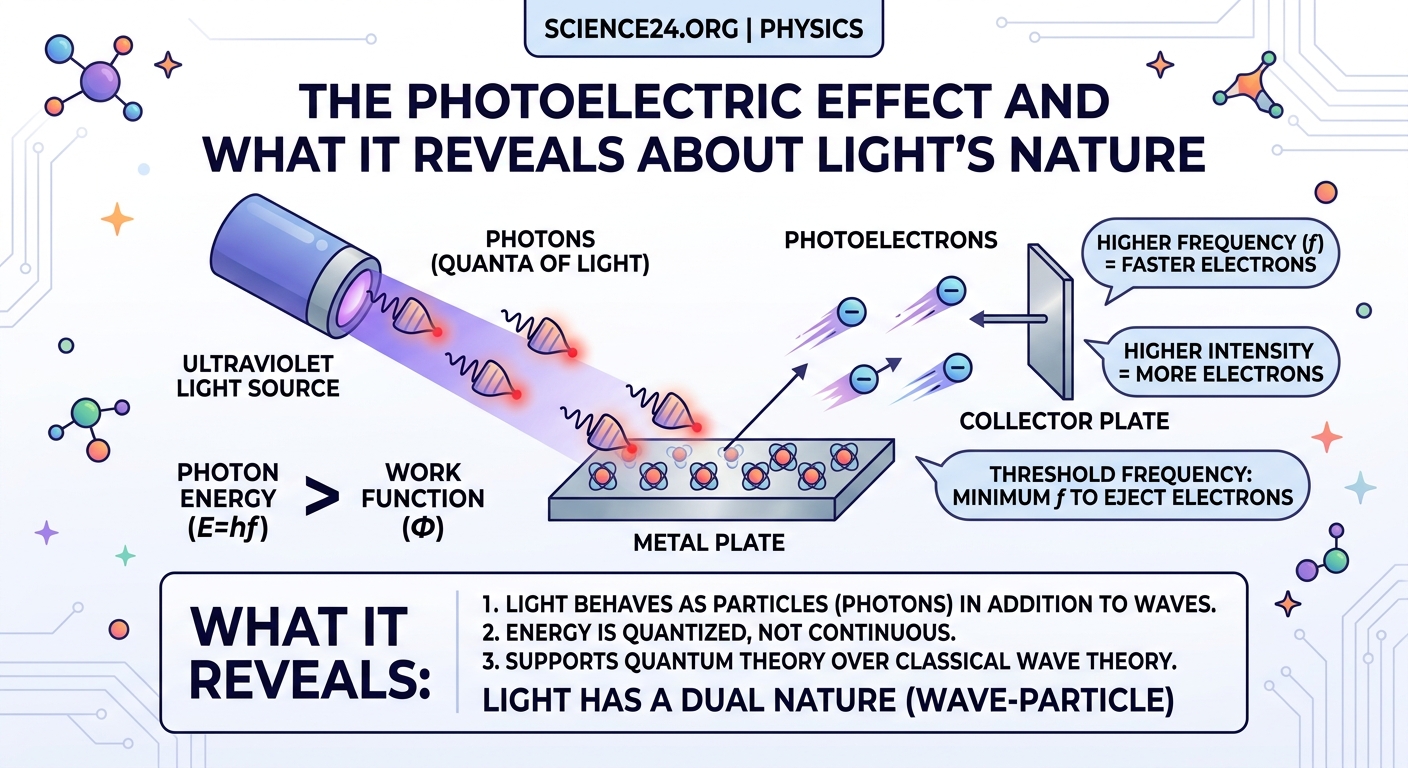
The photoelectric effect occurs when light shines on certain metal surfaces and causes them to emit electrons. Scientists first noticed this phenomenon in the late 1800s during experiments with metal plates and electrical circuits.
When ultraviolet light hit zinc plates, electrons flew off the metal surface. Researchers could detect these ejected electrons as an electric current. The setup seemed straightforward, but the results defied all predictions based on wave theory.
Classical physics predicted that brighter light should always produce more energetic electrons. After all, bigger waves carry more energy. Yet experiments showed something different entirely.
Dim violet light ejected electrons immediately. Bright red light produced nothing, no matter how intense the beam became. The color of light mattered more than its brightness.
This observation contradicted everything physicists understood about electromagnetic radiation at the time.
Why wave theory failed to explain the observations
Wave theory made specific predictions about how light should interact with metal surfaces. Every prediction turned out wrong.
According to wave theory, electrons should absorb energy gradually from the incoming light waves. Brighter light meant bigger waves carrying more energy. Given enough time, even dim light should eventually provide enough energy to free electrons from the metal.
The theory also predicted that increasing light intensity should produce faster electrons. More wave energy should translate directly into more kinetic energy for the ejected particles.
Reality disagreed on all counts.
Experiments revealed three key observations that wave theory could not explain:
- Electron ejection happened instantly, with no delay time for energy accumulation
- Below a certain light frequency, no electrons ejected regardless of intensity
- Above that threshold frequency, even dim light produced immediate electron emission
- Electron energy depended only on light frequency, not intensity
The threshold frequency concept made no sense within wave theory. Why would red light fail completely while violet light succeeded immediately? Waves should deliver energy continuously regardless of color.
Einstein’s revolutionary photon explanation
Einstein proposed a radical solution in 1905. Light does not travel as continuous waves. Instead, it consists of discrete energy packets called photons.
Each photon carries a specific amount of energy determined by the light’s frequency. The relationship follows a simple equation:
E = hf
Where E represents photon energy, h is Planck’s constant (6.626 × 10⁻³⁴ J·s), and f indicates frequency.
Higher frequency means more energetic photons. Violet light photons pack more punch than red light photons. This explains why color matters more than brightness.
When a photon strikes an electron in the metal, it transfers all its energy in a single interaction. The electron either receives enough energy to escape or it doesn’t. There is no gradual buildup.
This particle model explained every puzzling observation perfectly.
For an electron to escape the metal surface, it must overcome the work function, the binding energy holding it in place. Different metals have different work functions. Zinc requires less energy than platinum.
If a photon’s energy exceeds the work function, the electron escapes immediately. Any leftover energy becomes the electron’s kinetic energy. If the photon energy falls short, nothing happens, even with billions of photons hitting the surface.
The mathematical relationship looks like this:
KE = hf – φ
Where KE represents the electron’s kinetic energy and φ (phi) represents the work function.
How to understand threshold frequency
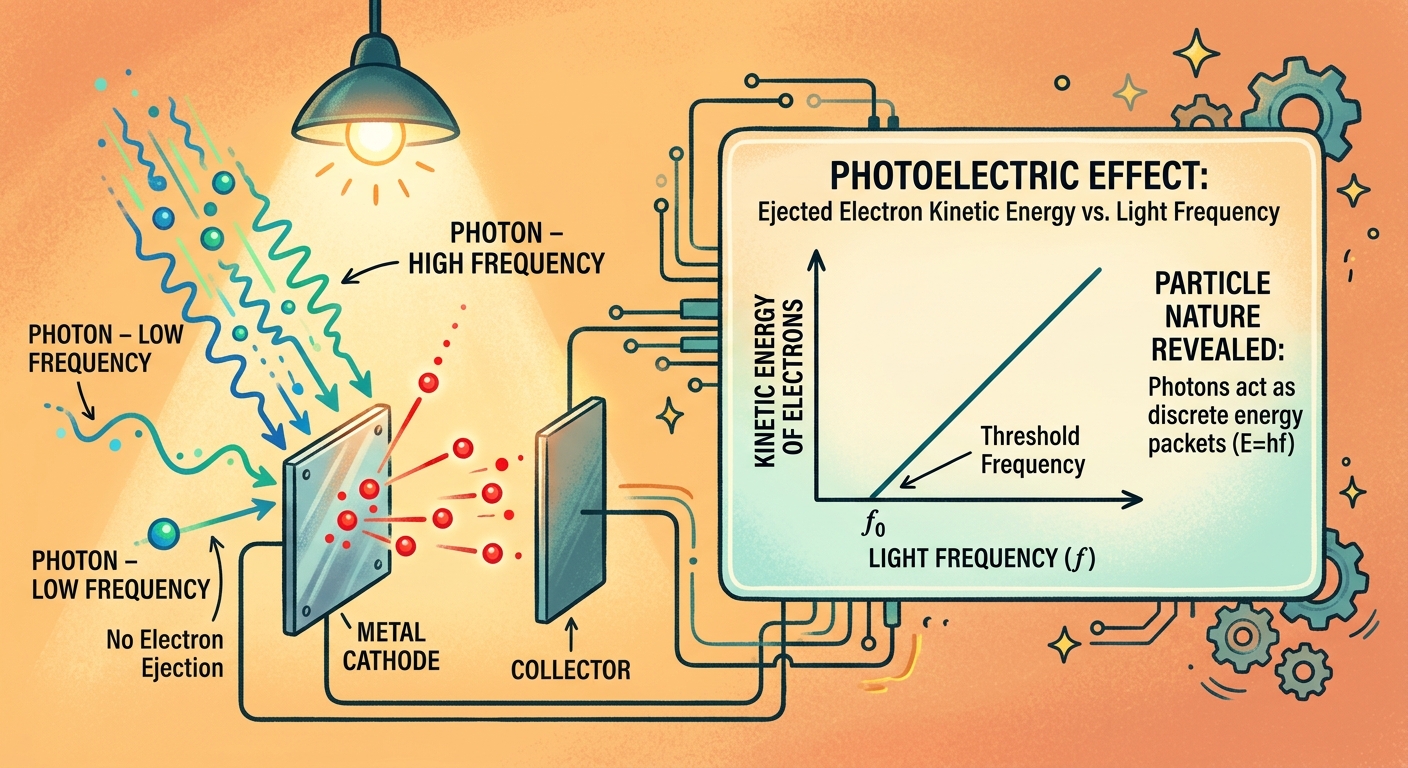
Every metal has a minimum frequency of light required to eject electrons. This threshold frequency (f₀) depends entirely on the work function.
At the threshold frequency, photon energy exactly equals the work function. The equation becomes:
hf₀ = φ
Below this frequency, photons lack sufficient energy. You could shine a trillion red photons per second on zinc, and not one electron would escape. Each individual photon simply doesn’t carry enough energy to do the job.
Above the threshold frequency, electrons eject immediately. The excess energy beyond the work function converts to kinetic energy, making the electrons move faster.
| Light Property | Effect on Electron Ejection | Effect on Electron Energy |
|---|---|---|
| Frequency above threshold | Electrons eject | Higher frequency = faster electrons |
| Frequency below threshold | No ejection occurs | No electrons produced |
| High intensity above threshold | More electrons eject | No change in individual electron speed |
| Low intensity above threshold | Fewer electrons eject | No change in individual electron speed |
This table clarifies a common point of confusion. Intensity affects the number of electrons ejected because more photons hit the surface per second. However, each electron’s energy depends only on the individual photon frequency that freed it.
Practical steps to solve photoelectric effect problems
Physics courses frequently test understanding through calculation problems. Here’s a systematic approach that works reliably:
-
Identify the given information and convert all units to standard SI units (frequency in Hz, energy in joules or electron volts).
-
Determine what the problem asks for: threshold frequency, work function, maximum kinetic energy, or stopping potential.
-
Select the appropriate equation based on what you need to find and what information you have.
-
Solve algebraically before inserting numbers to catch mistakes early.
-
Check that your answer makes physical sense given the context of the problem.
For example, if you calculate a work function larger than the photon energy, you know immediately that electrons cannot eject. If you find negative kinetic energy, you’ve made an error somewhere.
Many students struggle with unit conversions between joules and electron volts. Remember that 1 eV = 1.602 × 10⁻¹⁹ J. Photon energies often appear in eV because the numbers are more manageable.
Common misconceptions students should avoid
Several persistent misunderstandings plague students learning about the photoelectric effect. Recognizing these helps build accurate mental models.
The biggest mistake is thinking brighter light always means more energetic electrons. Brightness relates to photon quantity, not individual photon energy. A million low-energy photons cannot combine their energies to free a single electron. Each photon-electron interaction is independent.
Another confusion involves the work function. Students sometimes think it represents the total energy needed to remove all electrons from a metal sample. Actually, it’s the minimum energy required to remove one electron from the surface. Electrons deeper in the metal require even more energy.
Some learners expect a delay between light exposure and electron emission, reasoning that electrons need time to absorb enough energy. This expectation comes from wave thinking. Photon interactions happen essentially instantaneously, typically within nanoseconds.
The relationship between stopping potential and kinetic energy also causes trouble. Stopping potential is the voltage needed to prevent the most energetic electrons from reaching a detector. It relates directly to maximum kinetic energy through:
eV₀ = KE_max
Where e is the elementary charge and V₀ is the stopping potential.
Understanding the photoelectric effect requires abandoning classical intuitions about energy transfer. Light delivers energy in discrete chunks, not continuous streams. Each photon either has enough energy to do the job or it doesn’t. There’s no middle ground, no gradual accumulation. This all-or-nothing character defines quantum behavior.
Wave-particle duality emerges from the evidence
The photoelectric effect forced physicists to accept that light exhibits both wave and particle properties. This concept, called wave-particle duality, seems contradictory but accurately describes reality.
Light demonstrates wave properties through interference and diffraction patterns. When light passes through narrow slits, it creates characteristic patterns that only waves can produce. These experiments prove light spreads through space as electromagnetic oscillations.
Yet the photoelectric effect proves light also behaves as discrete particles during interactions with matter. Photons carry specific energy quanta and transfer that energy in individual packets.
Both descriptions are correct. Light is neither purely wave nor purely particle. It’s something more complex that exhibits different aspects depending on how you observe it.
This duality extends beyond light. Electrons, protons, and all matter also show wave-particle behavior. The connection between why objects fall at the same rate regardless of mass and quantum mechanics runs deeper than classical physics ever suggested.
The photoelectric effect opened the door to quantum mechanics by proving that energy transfers happen in discrete amounts. This quantization principle revolutionized physics and led to technologies from solar panels to digital cameras.
Applications that depend on photon behavior
Modern technology relies heavily on the photoelectric effect. Understanding light’s particle nature enabled inventions that shape daily life.
Solar panels convert sunlight directly into electricity using the photoelectric effect. Photons strike semiconductor materials and eject electrons, creating current flow. The efficiency depends on matching the semiconductor’s band gap to the solar spectrum’s photon energies.
Digital camera sensors use photoelectric materials to detect light. Each pixel contains a photodiode that releases electrons when photons hit it. The number of electrons determines the pixel’s brightness value. This technology replaced chemical film because it converts light to electrical signals instantly and reliably.
Night vision devices amplify dim light through photoelectric multiplication. Incoming photons strike a photocathode, ejecting electrons. These electrons accelerate through a voltage and strike a phosphor screen, producing visible light. A single photon can trigger thousands of output photons.
Photomultiplier tubes in scientific instruments detect extremely weak light signals. They work similarly to night vision but with even greater amplification. These devices enable experiments in particle physics, astronomy, and medical imaging.
The stopping potential measurements used in photoelectric experiments led directly to techniques for determining Planck’s constant with high precision. This fundamental constant appears throughout quantum mechanics and relates energy to frequency for all particles.
Experimental verification you can understand
The classic photoelectric effect experiment uses surprisingly simple equipment. Understanding the setup clarifies how scientists confirmed Einstein’s photon model.
A vacuum tube contains two electrodes: an emitter (photocathode) and a collector. Light shines on the photocathode through a quartz window. When electrons eject, they travel toward the collector, creating measurable current.
A variable voltage source between the electrodes allows researchers to either help or hinder electron flow. Positive voltage on the collector pulls electrons toward it. Negative voltage pushes them back toward the emitter.
By making the collector increasingly negative, experimenters find the stopping potential where current drops to zero. This voltage reveals the maximum kinetic energy of ejected electrons.
Researchers vary the light frequency using filters or a monochromator and measure the stopping potential for each frequency. Plotting stopping potential against frequency produces a straight line. The slope equals h/e (Planck’s constant divided by elementary charge). The x-intercept gives the threshold frequency.
This linear relationship provided powerful evidence for Einstein’s photon model. Wave theory predicted a completely different curve shape.
Modern versions of this experiment appear in many undergraduate physics labs. Students can personally verify the fundamental principles of quantum mechanics using equipment that fits on a lab bench, similar to how understanding heat transfer through conduction, convection, and radiation becomes clearer through hands-on observation.
Mathematical relationships you need to master
Success with photoelectric effect problems requires comfort with several key equations. Each formula connects specific physical quantities.
The fundamental photon energy equation:
E = hf = hc/λ
This relates photon energy to either frequency (f) or wavelength (λ). Since c = λf for electromagnetic radiation, you can use whichever form fits your given information. Higher frequency means shorter wavelength and more energy.
The photoelectric equation:
KE_max = hf – φ
This gives the maximum kinetic energy of ejected electrons. Only electrons at the metal surface with minimum binding energy achieve this maximum. Electrons from deeper layers have less kinetic energy after escaping.
The threshold frequency relationship:
f₀ = φ/h
This determines the minimum light frequency needed for electron ejection. Below this frequency, photon energy cannot overcome the work function.
The stopping potential equation:
eV₀ = KE_max = hf – φ
This connects electrical measurements to kinetic energy. Stopping potential provides an experimental method to determine electron energy without directly measuring velocity.
When working problems, always check units carefully. Frequency uses hertz (Hz), energy uses joules (J) or electron volts (eV), and wavelength uses meters (m). Converting between these requires attention to detail that separates correct answers from careless errors.
Historical context that shaped quantum mechanics
The photoelectric effect emerged during a crisis in physics. Classical theories worked beautifully for most phenomena but failed spectacularly for others.
Max Planck introduced the quantum concept in 1900 while studying blackbody radiation. He proposed that electromagnetic energy could only be emitted or absorbed in discrete amounts proportional to frequency. This solved the ultraviolet catastrophe problem but seemed like a mathematical trick rather than physical reality.
Einstein took Planck’s idea seriously in his 1905 paper on the photoelectric effect. He argued that light itself consists of quanta, not just the emission and absorption processes. This bold claim went beyond Planck’s original proposal.
The physics community resisted Einstein’s photon concept for years. Even after experimental verification, many prominent physicists preferred to think of light as waves with some quantum restrictions. The particle interpretation seemed too radical.
Robert Millikan spent a decade performing meticulous experiments trying to disprove Einstein’s photoelectric equation. Instead, his precise measurements confirmed every prediction. Millikan won the Nobel Prize in 1923 partly for this work, despite his initial skepticism.
Einstein received his Nobel Prize in 1921 specifically for explaining the photoelectric effect, not for his more famous relativity theories. The prize committee considered the photoelectric work more solidly established at the time.
This historical development shows how scientific understanding progresses through both theoretical insight and experimental verification. Bold ideas need careful testing before acceptance, even when they come from brilliant minds.
Connecting photons to broader quantum principles
The photoelectric effect represents just one piece of the quantum mechanics puzzle. Its implications extend far beyond metal surfaces and ejected electrons.
Photons demonstrate that energy quantization is fundamental to nature, not just a mathematical convenience. All electromagnetic radiation, from radio waves to gamma rays, consists of photons with energies determined by frequency.
The concept of wave-particle duality applies universally. Louis de Broglie proposed in 1924 that matter particles also have wave properties. His hypothesis proved correct, leading to electron microscopes and other technologies exploiting matter waves.
The probabilistic nature of quantum mechanics connects to the photoelectric effect through the randomness of individual photon interactions. You cannot predict which specific electron a photon will strike or exactly when emission occurs. You can only calculate probabilities for large numbers of events.
Energy conservation remains absolute in quantum mechanics, but it applies to individual photon-electron interactions. Each collision conserves energy and momentum, even though the outcomes vary randomly within quantum constraints.
The photoelectric effect also illustrates the measurement problem in quantum mechanics. Before detection, an electron exists in a superposition of states. The act of measurement, whether hitting a detector or being stopped by an electric field, collapses this superposition to a definite outcome, much like how understanding imaginary numbers without the confusion requires accepting mathematical concepts that seem impossible at first.
Studying strategies for exam success
Mastering the photoelectric effect for exams requires both conceptual understanding and problem-solving practice. Different question types demand different preparation approaches.
Conceptual questions test whether you truly understand photon behavior versus wave behavior. Practice explaining why intensity affects electron quantity but not individual electron energy. Describe why threshold frequency exists and what it means physically. These explanations should use everyday language without sacrificing accuracy.
Calculation problems require fluency with the key equations. Work through varied examples changing which quantity you solve for. Sometimes you’ll calculate work function from threshold frequency. Other times you’ll find maximum kinetic energy given photon wavelength and work function. Flexibility comes from repeated practice.
Graph interpretation questions appear frequently. You might see stopping potential versus frequency, current versus intensity, or kinetic energy versus wavelength. Understanding the physical meaning behind each graph’s shape and intercepts proves your grasp of underlying principles.
Comparison questions ask you to predict how changing one variable affects others. If frequency increases while intensity stays constant, what happens to electron count and energy? Building a mental model of the photon-electron interaction process makes these predictions straightforward.
Create a summary sheet listing all key equations, their variables, and when to use each one. Include the values of fundamental constants like Planck’s constant and elementary charge. This reference sheet helps during problem-solving practice and serves as a study guide.
Work problems without looking at solutions first. Struggling productively builds stronger understanding than passively reading worked examples. Check your approach only after attempting the problem yourself, similar to strategies in the 72-hour study plan that actually works for chemistry exams.
How this discovery transformed our understanding
The photoelectric effect fundamentally changed how physicists think about reality. Before Einstein’s explanation, energy seemed continuous and infinitely divisible. Afterward, the quantum nature of energy became undeniable.
This shift in perspective enabled the entire field of quantum mechanics. Niels Bohr used quantized energy levels to explain atomic spectra. Erwin Schrödinger developed wave mechanics to describe electron behavior in atoms. Werner Heisenberg formulated matrix mechanics and the uncertainty principle. All these developments built on the foundation that energy comes in discrete packets.
The technological impact extends beyond obvious applications like solar panels. Semiconductor physics relies completely on quantum mechanics. Every computer chip, LED, laser, and transistor exists because engineers understand how electrons and photons interact at the quantum level.
Medical imaging techniques from PET scans to radiation therapy depend on controlling photon and particle energies with quantum precision. Materials science uses quantum principles to design everything from superconductors to quantum dots.
The photoelectric effect proved that nature operates fundamentally differently at small scales than our everyday experience suggests. This realization ranks among the most important scientific discoveries ever made.
Light carries its nature in every interaction. Each photon that strikes your eye, charges your phone, or powers a satellite demonstrates the same quantum principles that puzzled physicists over a century ago. The photoelectric effect revealed a deeper layer of reality that continues shaping technology and understanding today.
Making quantum concepts stick
Grasping the photoelectric effect means accepting that light behaves in ways that feel wrong initially. Your intuition developed in a world of continuous processes and gradual changes. Quantum mechanics operates differently.
Think of photons as indivisible currency units. You cannot split a penny into smaller monetary amounts for everyday transactions. Similarly, you cannot split a photon into smaller energy units during interactions. The photon delivers all its energy or none of it.
This discrete nature explains why certain phenomena happen instantly rather than gradually. Electron ejection occurs immediately when photon energy exceeds the work function because the entire energy transfer happens in a single event. There’s no accumulation period because there’s nothing to accumulate between whole photons.
The wave-particle duality makes sense when you stop trying to force light into familiar categories. Light isn’t “really” a wave that sometimes acts like a particle, or vice versa. It’s a quantum object that exhibits wave properties in some experiments and particle properties in others. Both aspects are equally real and fundamental.
Your growing understanding of quantum mechanics will serve you well beyond this single topic. The same principles apply throughout modern physics, chemistry, and engineering. The photoelectric effect provides a clear, experimentally verifiable entry point into this strange and fascinating realm where classical intuition fails but mathematical description succeeds beautifully.




