Whether you’re holding a warm coffee mug, feeling the sun on your face, or watching steam rise from a pot, you’re witnessing heat transfer in action. Understanding how thermal energy moves from one place to another isn’t just an academic exercise. It’s the foundation for everything from designing efficient buildings to understanding weather patterns and even cooking your dinner properly.
Heat transfers through three distinct mechanisms: conduction (direct contact between materials), convection (movement through fluids like air or water), and radiation (electromagnetic waves requiring no medium). Each method operates under different principles and appears in countless everyday situations, from cooking food to regulating body temperature. Mastering these concepts helps you understand thermal physics and solve practical engineering problems.
What Makes Heat Transfer Different From Temperature
Temperature measures how hot or cold something is. Heat transfer describes the actual movement of thermal energy between objects or areas.
This distinction matters because two objects at the same temperature don’t exchange heat. Only when a temperature difference exists does thermal energy flow from the warmer region to the cooler one.
The rate of this energy flow depends on several factors. Material properties, surface area, temperature difference, and the specific transfer mechanism all play roles.
Conduction Moves Heat Through Direct Contact
Conduction happens when thermal energy passes through a material or between materials touching each other. Atoms and molecules vibrate faster when heated, and this vibration spreads to neighboring particles.
Think of a metal spoon sitting in hot soup. The handle gradually warms up even though it’s not submerged. Heat travels along the spoon through molecular collisions.
Metals conduct heat exceptionally well because they contain free electrons that move easily through their structure. These electrons carry thermal energy much faster than molecular vibrations alone could manage.
Wood, plastic, and air conduct heat poorly. We call these materials insulators. Your winter coat works because it traps air, which prevents heat from conducting away from your body.
The Math Behind Conduction
Fourier’s Law describes how heat flows through conduction:
Q = kA(T₁ – T₂)/d
Where:
– Q represents heat flow rate (watts)
– k is thermal conductivity (material property)
– A is cross-sectional area
– T₁ and T₂ are temperatures at each end
– d is thickness or distance
Higher thermal conductivity means faster heat transfer. Copper has a k value around 400 W/(m·K), while wood sits near 0.1 W/(m·K).
This equation explains why thick walls insulate better than thin ones. Doubling the thickness halves the heat flow rate, assuming everything else stays constant.
Practical Conduction Examples
Here are situations where conduction dominates:
- Cooking with a cast iron skillet that distributes heat evenly across the cooking surface
- Touching a cold metal railing in winter and feeling heat drain from your hand
- Using a heating pad where warmth transfers directly to sore muscles
- Feeling cold tile floors that conduct heat away from your bare feet
Building designers use conduction principles when selecting insulation materials. Double-pane windows trap air between glass layers, reducing conductive heat loss dramatically compared to single-pane designs.
Convection Requires Fluid Movement
Convection transfers heat through the bulk movement of fluids (liquids or gases). Unlike conduction, which relies on stationary particles passing energy, convection physically moves warm fluid to cooler areas.
Two types exist: natural convection and forced convection.
Natural convection occurs when density differences cause fluid movement. Warm air rises because it becomes less dense as it heats up. Cool air sinks to replace it, creating a circulation pattern called a convection current.
Forced convection uses external forces like fans or pumps to move fluid. Your car’s cooling system pumps coolant through the engine block, carrying away heat much faster than natural convection could.
How Convection Works in Your Home
Room heating demonstrates convection beautifully. A radiator or baseboard heater warms nearby air. That air expands, becomes lighter, and rises toward the ceiling. Cooler air flows in to replace it, gets heated, and the cycle continues.
Ceiling fans don’t actually cool air. They push warm air down in winter or create a breeze in summer that helps sweat evaporate. Both applications manipulate convection currents.
Ocean currents represent massive convection systems. Warm water near the equator flows toward the poles while cold water sinks and returns along the ocean floor. These currents regulate global climate patterns.
Calculating Convection Heat Transfer
Newton’s Law of Cooling governs convection:
Q = hA(T_surface – T_fluid)
Where:
– Q is heat transfer rate
– h is the convection coefficient
– A is surface area
– T_surface is the object’s surface temperature
– T_fluid is the surrounding fluid temperature
The convection coefficient h depends on fluid properties, flow velocity, and surface geometry. Moving air has a higher h value than still air, which is why windy winter days feel colder.
Radiation Needs No Medium At All
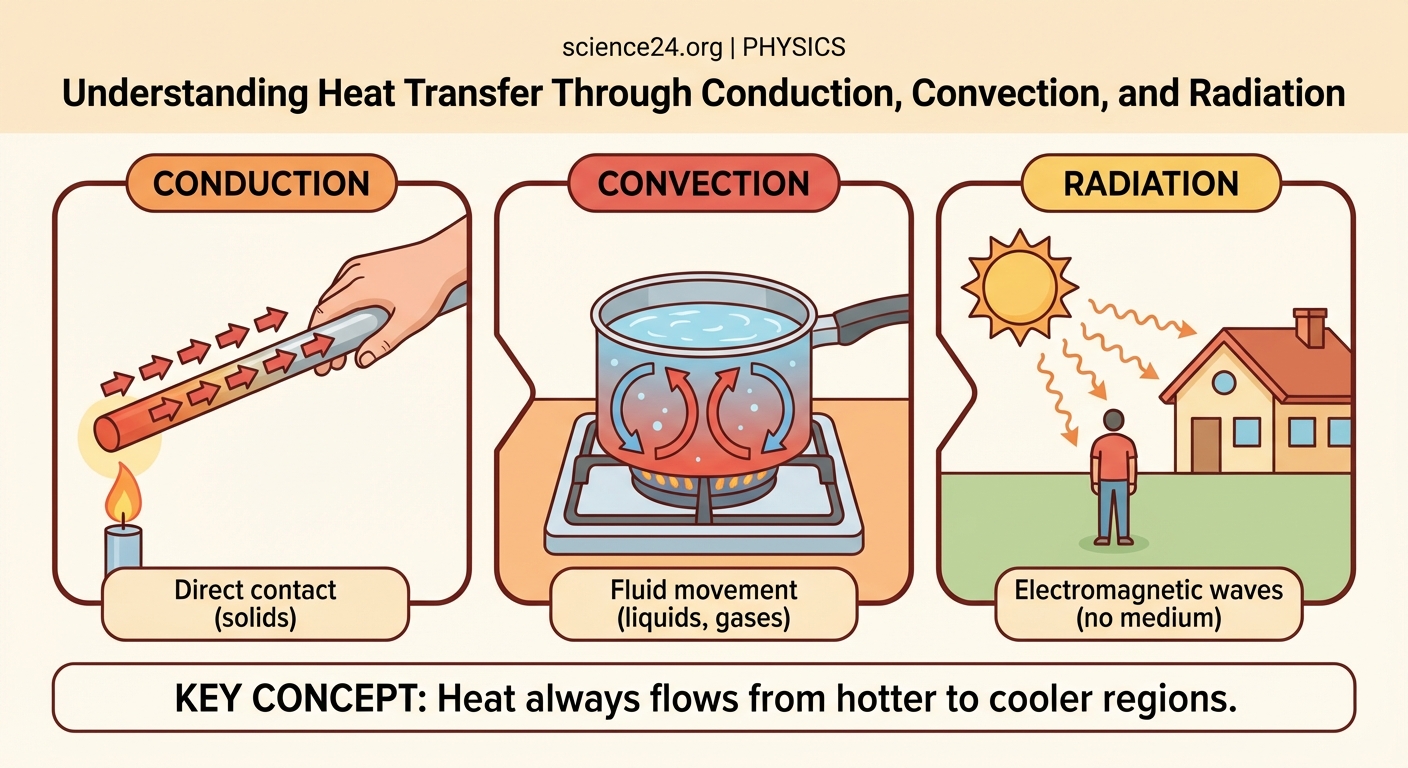
Radiation transfers heat through electromagnetic waves. Unlike conduction and convection, radiation requires no physical matter between the heat source and receiver.
The sun warms Earth across 93 million miles of mostly empty space. No conduction or convection could bridge that gap, but radiation crosses it easily.
All objects emit thermal radiation based on their temperature. Hotter objects radiate more energy and at shorter wavelengths. This explains why heating elements glow red, then orange, then white as they get hotter.
Understanding the Stefan-Boltzmann Law
The Stefan-Boltzmann Law quantifies radiation:
Q = εσA(T⁴)
Where:
– Q is radiated power
– ε is emissivity (0 to 1)
– σ is the Stefan-Boltzmann constant (5.67 × 10⁻⁸ W/(m²·K⁴))
– A is surface area
– T is absolute temperature in Kelvin
Notice the T⁴ term. Doubling absolute temperature increases radiation by 16 times. This makes radiation the dominant heat transfer mode at high temperatures.
Emissivity describes how effectively a surface radiates. Perfect black bodies have ε = 1. Shiny metallic surfaces have low emissivity, which is why thermos bottles use reflective coatings to minimize heat loss.
Radiation in Daily Life
Radiation surrounds you constantly:
- Feeling warmth from a fireplace even when you’re not touching anything
- Using a microwave that generates electromagnetic waves to heat food molecules
- Wearing white clothing in summer because light colors reflect more radiation than dark ones
- Sitting under an infrared heat lamp at a restaurant patio
Greenhouse gases trap radiation in Earth’s atmosphere. Carbon dioxide and methane absorb infrared radiation that Earth emits, then re-radiate it in all directions, including back toward the surface. This process drives climate warming.
Comparing All Three Mechanisms Side by Side
| Method | Requires Matter? | Dominant In | Speed | Example |
|---|---|---|---|---|
| Conduction | Yes (solid contact) | Solids, stationary fluids | Slow to moderate | Metal spoon heating |
| Convection | Yes (moving fluid) | Liquids and gases | Moderate | Boiling water |
| Radiation | No | All states, especially across space | Speed of light | Sunlight warming skin |
Most real situations involve multiple mechanisms working simultaneously. When you boil water, conduction heats the pot bottom, convection circulates hot water upward, and radiation from the burner contributes additional energy.
Understanding which mechanism dominates helps solve problems efficiently. For instance, improving home insulation focuses on reducing conduction through walls while minimizing convection through air gaps.
Common Misconceptions About Heat Transfer
Students often confuse heat with temperature. Remember: temperature is a measure, heat is energy in transit.
Another mistake involves thinking cold transfers. Cold isn’t a substance that flows. Only heat moves, always from hot to cold. When you touch ice, heat leaves your hand rather than cold entering it.
Some people believe thicker materials always insulate better. While thickness helps with conduction, it doesn’t prevent radiation. Space blankets use thin reflective material to block radiative heat loss effectively.
The concept that metals feel colder than wood at room temperature puzzles many. Both are actually the same temperature. Metal conducts heat away from your hand faster, creating a sensation of coldness. Wood’s poor conductivity means heat doesn’t leave your skin as rapidly.
Solving Heat Transfer Problems Step by Step
When tackling physics problems or engineering calculations, follow this process:
- Identify which heat transfer mechanism(s) apply to your situation
- Write down known values including temperatures, dimensions, and material properties
- Select the appropriate equation (Fourier’s Law, Newton’s Law, or Stefan-Boltzmann)
- Convert all units to a consistent system (usually SI units)
- Solve for the unknown variable and check if the answer makes physical sense
“The key to mastering thermodynamics is recognizing that heat always flows downhill on the temperature scale. Every heat transfer problem becomes manageable once you identify the hot source, cold sink, and pathway between them.” (Physics education research)
This systematic approach works whether you’re calculating heat loss through a building wall or determining cooking times for different materials. Just like mastering calculation techniques builds mathematical confidence, practicing heat transfer problems builds physical intuition.
Real-World Applications Across Industries
Engineers apply heat transfer principles constantly. Computer chip designers must remove heat faster than processors generate it. They use conduction through heat sinks, convection from fans, and sometimes liquid cooling systems.
Automotive engineers balance engine cooling needs with aerodynamic efficiency. Radiators use all three mechanisms: conduction from engine to coolant, convection as coolant circulates, and radiation from the radiator surface.
Climate control systems in buildings optimize energy use by managing heat transfer. Proper insulation reduces conduction, sealed windows minimize convection, and reflective coatings limit radiation.
Food scientists understand that different cooking methods emphasize different transfer modes:
- Baking uses convection (hot air) and radiation (oven walls)
- Frying relies primarily on conduction (pan contact)
- Grilling combines conduction (grill grates) with radiation (hot coals or elements)
- Microwaving uses radiation at specific frequencies
Measuring and Testing Heat Transfer
Laboratory experiments help students grasp these concepts concretely. Simple tests include:
Testing conductivity: Place one end of different materials in hot water and measure how long heat takes to reach the other end. Metals transfer heat fastest, followed by ceramics, then plastics and wood.
Observing convection: Add food coloring to water in a clear container, then heat one side. Watch colored currents rise from the heat source and sink as they cool.
Demonstrating radiation: Hold your hand near (not touching) a lit incandescent bulb. You feel warmth from radiation. A compact fluorescent bulb at the same brightness feels cooler because it produces less infrared radiation.
These hands-on activities build intuition that equations alone can’t provide. Similar to how understanding abstract concepts requires both theory and practice, heat transfer becomes clear through experience.
Avoiding Common Calculation Errors
Students make predictable mistakes when working heat transfer problems. Recognizing these helps you avoid them:
- Unit inconsistency: Mixing Celsius with Kelvin, or watts with joules per second (they’re equivalent, but context matters)
- Forgetting area: Heat flow depends on surface area; larger areas transfer more heat
- Ignoring composite materials: Walls contain multiple layers with different conductivities
- Misapplying steady-state equations: Some formulas assume equilibrium conditions that don’t always exist
Creating a reference sheet with formulas, units, and typical values helps during exams. Include thermal conductivities for common materials, emissivity values, and unit conversion factors.
Double-checking answers against physical intuition catches many errors. If your calculation suggests a wooden spoon conducts heat better than copper, something went wrong.
Connecting Heat Transfer to Other Physics Topics
Thermodynamics and heat transfer intertwine closely. The second law of thermodynamics states that heat naturally flows from hot to cold, never the reverse without external work. This principle underlies all three transfer mechanisms.
Energy conservation appears throughout heat transfer problems. Energy leaving one object must equal energy entering another (plus any lost to surroundings). This is similar to how energy behaves during collisions in mechanics.
Electromagnetic radiation connects heat transfer to wave physics. Infrared radiation, visible light, and radio waves all belong to the electromagnetic spectrum. Understanding wave properties helps explain why different surfaces absorb or reflect thermal radiation differently.
Statistical mechanics explains conduction at the molecular level. Temperature relates to average kinetic energy of particles. When fast-moving molecules collide with slower ones, kinetic energy transfers, which we observe as heat conduction.
Mastering Heat Transfer for Exams and Beyond
Success in thermodynamics courses requires understanding concepts, not just memorizing formulas. Ask yourself why each mechanism works the way it does.
Practice problems covering all three mechanisms. Start with simple single-mechanism problems, then progress to combined scenarios that require analyzing multiple heat transfer modes simultaneously.
Draw diagrams showing heat flow direction and identify temperature gradients. Visual representations help organize your thinking and catch errors before they propagate through calculations.
Form study groups where members explain concepts to each other. Teaching someone else reveals gaps in your own understanding. When you can describe why hot air rises using density and buoyancy principles, you’ve truly grasped convection.
Create flashcards for material properties like thermal conductivity values, specific heat capacities, and emissivity coefficients. While you shouldn’t memorize every value, familiarity with typical ranges helps you estimate and verify answers.
Why These Three Mechanisms Matter Everywhere
Heat transfer isn’t confined to physics classrooms. It shapes weather patterns, influences architectural design, determines cooking outcomes, and drives technological innovation.
Understanding these principles helps you make better decisions. Should you buy a ceramic or stainless steel pan? Ceramic conducts heat more evenly. Which insulation works best for your attic? Materials with low thermal conductivity reduce conduction losses.
Climate science relies heavily on heat transfer models. Predicting global temperature changes requires understanding how radiation interacts with atmospheric gases, how ocean convection distributes heat, and how ice and snow reflect radiation back to space.
Medical applications include everything from fever management to surgical tools. Cryotherapy uses conduction to cool injured tissues. Radiation therapy targets cancer cells. Even your body’s cooling system through sweating depends on convection and evaporative heat transfer.
The next time you notice warmth or cold, pause and identify which mechanism is at work. That metal bench feeling cold in winter? Conduction pulling heat from your body. Steam rising from your coffee? Convection currents carrying heat upward. Sunshine warming your face through a window? Radiation crossing the glass barrier. Recognition builds intuition, and intuition makes complex problems manageable.

Leave a Reply