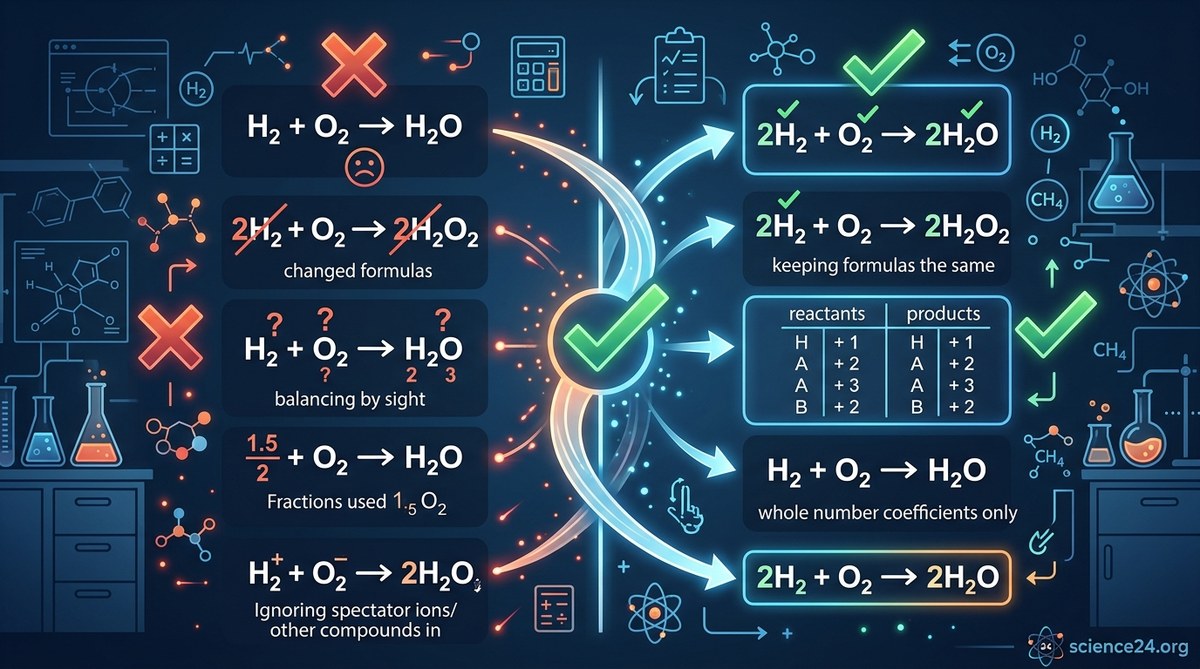
Balancing chemical equations feels like solving a puzzle where one wrong move ruins everything. You stare at the equation, add a coefficient here, adjust a subscript there, and suddenly nothing works. Your atoms don’t match. Your teacher marks it wrong. You wonder why something that looks so simple keeps tripping you up.
The truth is, most students make the same handful of errors when balancing equations. These mistakes aren’t about being bad at chemistry. They’re about missing key concepts that nobody explained clearly. Once you understand what’s going wrong and why, balancing equations becomes straightforward.
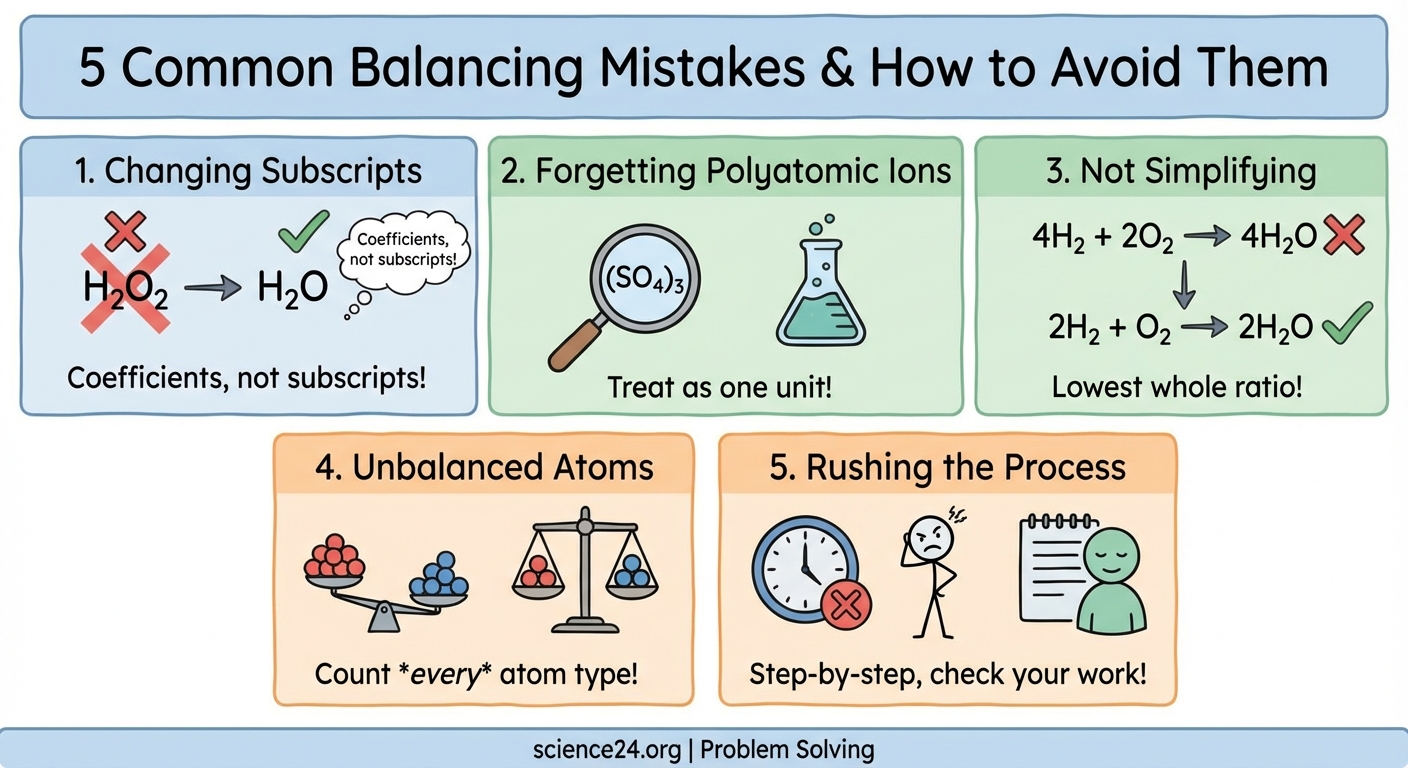
Students commonly fail to balance chemical equations because they change subscripts instead of coefficients, skip counting polyatomic ions as units, forget to simplify final coefficients, ignore the law of conservation of mass, or rush without checking their work. Understanding these five mistakes and learning systematic correction strategies will dramatically improve your equation balancing accuracy and chemistry exam performance.
Changing Subscripts Instead of Adding Coefficients
This mistake tops the list because it fundamentally breaks chemistry rules.
When you change a subscript, you create a completely different substance. Writing H₂O₂ instead of H₂O doesn’t just adjust the balance. It changes water into hydrogen peroxide. These are entirely different chemicals with different properties, different reactions, and different behaviors.
Subscripts tell you how atoms bond within a molecule. They’re locked in place by the chemical formula. You cannot touch them during balancing.
Coefficients, written in front of the entire formula, tell you how many molecules participate in the reaction. These are the only numbers you can adjust.
Correct approach:
- Write out the unbalanced equation with correct formulas.
- Count atoms of each element on both sides.
- Add coefficients in front of complete formulas only.
- Recount after each change.
- Repeat until balanced.
Consider this common example. Students trying to balance the combustion of methane often make this error:
| Wrong Approach | Why It Fails | Correct Approach |
|---|---|---|
| CH₄ + O₂ → CO₃ + H₂O | Changed CO₂ to CO₃, creating a nonexistent product | CH₄ + 2O₂ → CO₂ + 2H₂O |
| CH₄ + O₂ → CO₂ + H₃O | Changed H₂O to H₃O, creating hydronium ion instead of water | Uses coefficients to balance hydrogen |
| C₂H₄ + O₂ → CO₂ + H₂O | Changed CH₄ to C₂H₄, creating ethene instead of methane | Keeps original formulas intact |
The fix is simple but requires discipline. Before you write anything, confirm you have the correct chemical formulas for all reactants and products. Then promise yourself you’ll never touch those subscripts again.
“The chemical formula is sacred. Your job isn’t to create new molecules. Your job is to count how many of each molecule participates in the reaction.” — Chemistry teaching principle
Students who master this concept avoid roughly 40% of all balancing errors. Just like you wouldn’t change the rules of a game mid-play, you don’t change molecular formulas mid-balance.
Forgetting to Count Polyatomic Ions as Single Units
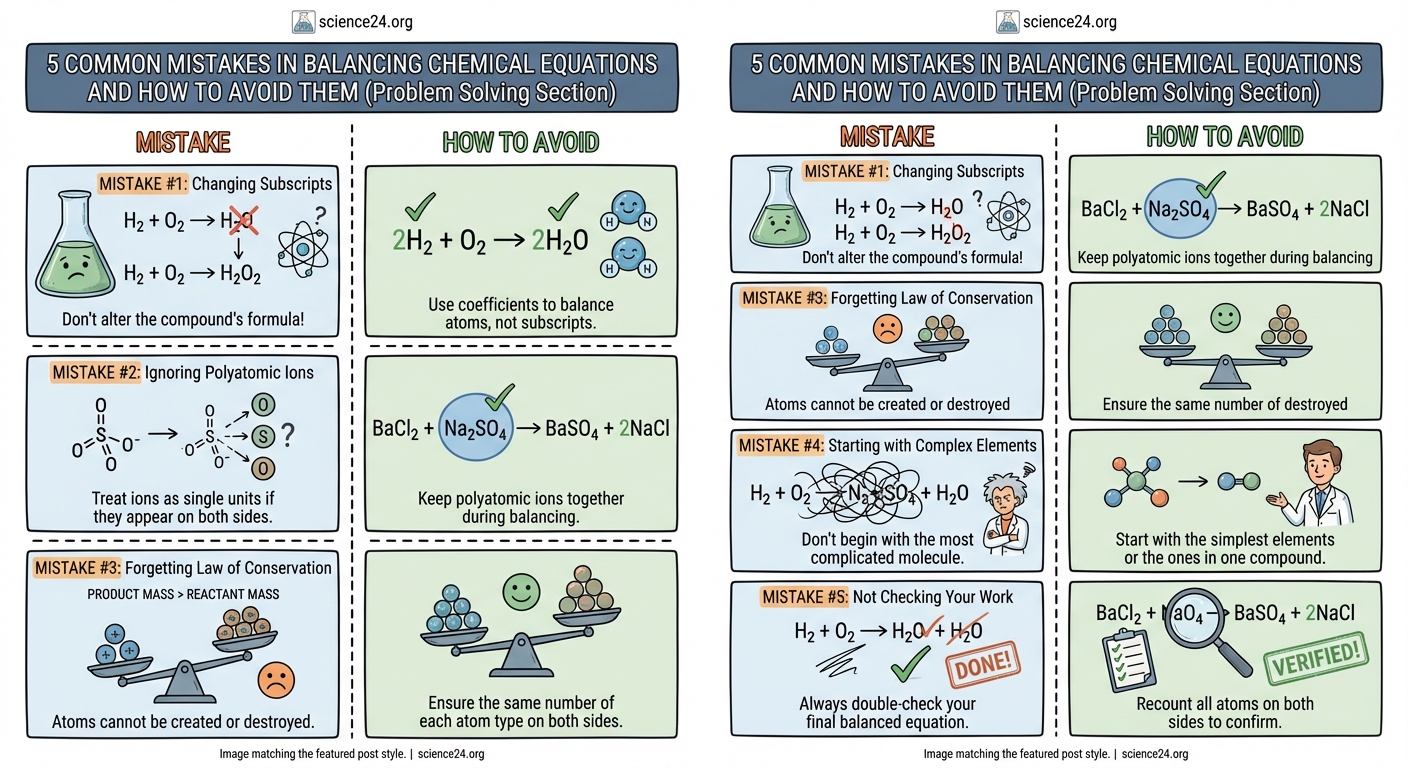
Polyatomic ions are groups of atoms that stick together and act as one unit during reactions.
Common examples include sulfate (SO₄²⁻), nitrate (NO₃⁻), carbonate (CO₃²⁻), and phosphate (PO₄³⁻). When these ions appear unchanged on both sides of an equation, you can balance them as complete units rather than counting individual atoms.
Many students waste time breaking these apart unnecessarily. They count sulfur atoms separately from oxygen atoms in sulfate, making the balancing process far more complicated than needed.
Here’s the efficient strategy:
- Scan your equation for polyatomic ions.
- Check if the same ion appears on both sides.
- If yes, treat the entire ion as one countable unit.
- Balance other elements first if the polyatomic ion causes confusion.
Example equation:
Ca(OH)₂ + H₃PO₄ → Ca₃(PO₄)₂ + H₂O
Notice that PO₄ appears on both sides. You can count phosphate groups instead of individual phosphorus and oxygen atoms.
Step-by-step balance:
- Calcium: 1 on left, 3 on right. Add coefficient 3 to Ca(OH)₂.
- Phosphate: 1 on left, 2 on right. Add coefficient 2 to H₃PO₄.
- Check hydrogen and oxygen separately now.
Final balanced equation: 3Ca(OH)₂ + 2H₃PO₄ → Ca₃(PO₄)₂ + 6H₂O
Students who break apart the phosphate groups often get lost in a maze of oxygen atoms. Those who recognize the pattern finish in half the time.
The same principle applies to hydroxide (OH⁻), ammonium (NH₄⁺), and other common polyatomic ions. Learning to spot these patterns makes balancing feel less like guessing and more like systematic problem solving, similar to how recognizing patterns helps with common algebra mistakes.
Skipping the Final Simplification Step
You’ve balanced your equation. All atoms match on both sides. You’re done, right?
Not always.
Many students submit answers like this:
4H₂ + 2O₂ → 4H₂O
Technically balanced, but not simplified. All coefficients share a common factor of 2. The simplest whole number ratio would be:
2H₂ + O₂ → 2H₂O
Chemistry convention requires the smallest possible whole number coefficients. Your teacher will mark the unsimplified version as incomplete or incorrect, even though the atom counts match.
This mistake appears most often when students:
- Balance oxygen or hydrogen first in combustion reactions.
- Use even numbers throughout to avoid fractions.
- Rush to finish without reviewing their final answer.
Quick simplification checklist:
- Look at all coefficients together.
- Find the greatest common divisor.
- Divide every coefficient by that number.
- Verify the equation still balances.
- Check that no coefficient equals 1 (which you can omit).
Some students deliberately use larger coefficients during the balancing process to avoid fractions. That’s perfectly fine as a working strategy. Just remember to simplify before writing your final answer.
Consider this progression:
- Working version: 6Na + 2AlCl₃ → 6NaCl + 2Al
- Simplified version: 3Na + AlCl₃ → 3NaCl + Al
Both balance correctly. Only the second one earns full credit.
One exception exists. If your equation includes a fraction that you cannot eliminate, multiply all coefficients by the denominator. For example, if you get:
C₃H₈ + (5/2)O₂ → 3CO₂ + 4H₂O
Multiply everything by 2:
2C₃H₈ + 5O₂ → 6CO₂ + 8H₂O
Fractions are acceptable as working steps but not in final answers.
Ignoring the Law of Conservation of Mass
Every chemical reaction follows one unbreakable rule. Atoms don’t appear or disappear. They just rearrange.
The number of each type of atom on the left side must exactly equal the number on the right side. No exceptions. No shortcuts. No creative accounting.
Yet students often:
- Balance one element while accidentally unbalancing another.
- Add atoms to make the math easier.
- Forget to recount after adding coefficients.
- Assume close enough is good enough.
Chemistry doesn’t grade on curves for atom counting. Either your equation respects conservation of mass or it doesn’t.
Systematic verification method:
- Create a two-column table.
- List every element present in the equation.
- Count atoms on the reactant side.
- Count atoms on the product side.
- Compare the columns.
- Adjust coefficients where numbers don’t match.
- Recount everything after each adjustment.
Example with the synthesis of ammonia:
N₂ + H₂ → NH₃ (unbalanced)
| Element | Reactants | Products | Balanced? |
|---|---|---|---|
| N | 2 | 1 | No |
| H | 2 | 3 | No |
After adding coefficients:
N₂ + 3H₂ → 2NH₃
| Element | Reactants | Products | Balanced? |
|---|---|---|---|
| N | 2 | 2 | Yes |
| H | 6 | 6 | Yes |
This table method catches errors that mental math misses. Students who skip this verification step often submit equations that look balanced but fail under careful inspection.
The law of conservation of mass isn’t just a chemistry principle. It’s a fundamental law of nature. Your equations must reflect reality. Matter cannot be created or destroyed in chemical reactions, only transformed, much like how energy behaves during collisions.
Rushing Without a Systematic Strategy
The final mistake isn’t about chemistry knowledge. It’s about process.
Students who struggle with balancing often jump around randomly. They adjust one coefficient, then another, then go back and change the first one again. They have no plan. They hope something will eventually work.
This approach wastes time and causes frustration. Balancing equations requires a methodical system.
Recommended balancing order:
- Balance elements that appear in only one compound on each side first.
- Save hydrogen and oxygen for last in combustion reactions.
- Balance polyatomic ions as units when possible.
- Handle the most complex molecule first.
- Leave single elements (like O₂ or H₂) for the end.
Why this order? Elements appearing in multiple compounds create cascading changes. Adjust them first, and you’ll redo work repeatedly. Single elements offer flexibility because changing their coefficient affects only one side of the equation.
Example combustion reaction:
C₃H₈ + O₂ → CO₂ + H₂O (unbalanced)
Step 1: Balance carbon (appears in one place on each side)
* 3 carbons on left, need 3 on right
* C₃H₈ + O₂ → 3CO₂ + H₂O
Step 2: Balance hydrogen (appears in one place on each side)
* 8 hydrogens on left, need 8 on right
* C₃H₈ + O₂ → 3CO₂ + 4H₂O
Step 3: Balance oxygen last (appears in multiple places on right)
* Right side: (3 × 2) + (4 × 1) = 10 oxygen atoms
* Need 10 on left: 5O₂
* C₃H₈ + 5O₂ → 3CO₂ + 4H₂O
Step 4: Verify all atoms balance
Step 5: Check if coefficients can be simplified (they cannot in this case)
Students who follow this system balance equations faster and with fewer errors. Those who skip around get stuck.
Another helpful technique involves writing atom counts above or below each element as you work. This visual tracking prevents losing your place.
Some reactions require algebraic approaches for complex balancing. In those cases, assign variables to coefficients and solve the system of equations. This advanced method appears less often in introductory courses but becomes valuable for redox reactions and other challenging scenarios.
The key insight is simple. Random guessing is not a strategy. Following a consistent process is.
Practice Techniques That Actually Work
Knowing what not to do only gets you halfway there. You also need effective practice methods.
Here’s what works:
- Start with simple synthesis and decomposition reactions before attempting combustion.
- Practice the same equation multiple times using different starting points.
- Time yourself to build speed without sacrificing accuracy.
- Work through equations without looking at answer keys first.
- Explain your process out loud to catch logical errors.
Create a personal error log. Every time you make a mistake, write down:
- The equation you were balancing
- What you did wrong
- Why it was wrong
- How to do it correctly
After a week, patterns emerge. You’ll notice if you consistently forget to simplify, or always mess up polyatomic ions, or rush through oxygen counting. Targeted practice on your specific weak points improves performance faster than random practice.
Many students benefit from working backward. Take a balanced equation and intentionally remove coefficients. Then rebalance it. This builds confidence because you know a solution exists.
Another powerful technique involves teaching someone else. When you explain the balancing process to a classmate or family member, you identify gaps in your own understanding. Questions they ask often reveal concepts you thought you knew but actually don’t.
Chemistry study groups work well for equation balancing because different students approach problems differently. Seeing multiple valid strategies expands your toolkit.
Digital tools and apps can provide immediate feedback, but don’t rely on them exclusively. Exams require you to balance equations by hand. Practice the way you’ll be tested.
Connecting Balancing Skills to Broader Chemistry Success
Balancing equations isn’t just a standalone skill. It connects to nearly every other chemistry topic you’ll encounter.
Stoichiometry calculations require balanced equations as starting points. You cannot calculate reactant amounts, limiting reagents, or percent yields without proper coefficients. One balancing error at the beginning ruins every calculation that follows.
Understanding chemical bonding helps you write correct formulas, which makes balancing easier. Recognizing exothermic reactions helps you predict products, which affects what you balance.
Students who struggle with balancing often have gaps in earlier concepts. If you’re having persistent trouble, review:
- How to write chemical formulas from names
- How to determine charges on ions
- How to recognize common polyatomic ions
- How to read the periodic table
These foundational skills make balancing feel natural rather than forced.
The mental discipline required for balancing equations also transfers to other STEM subjects. The systematic approach, careful verification, and attention to detail apply equally to solving physics problems, working through calculus, or debugging computer code.
Building Confidence Through Consistent Practice
You won’t master equation balancing overnight. Nobody does.
The students who excel aren’t naturally gifted. They simply practice more systematically and learn from their mistakes faster. They recognize patterns. They develop intuition about which coefficients to try first. They build a mental library of common reaction types.
Start small. Balance five simple equations correctly before moving to harder ones. Celebrate getting them right. Understand why you got them wrong when you make mistakes.
Your chemistry course will present hundreds of equations throughout the semester. Each one is practice. Each one builds skill. The equations that seem impossible now will feel routine after you’ve balanced fifty of them.
The five mistakes covered here account for most balancing errors students make. Avoid changing subscripts. Count polyatomic ions as units. Simplify your final answer. Respect conservation of mass. Follow a systematic process. Master these principles and balancing equations transforms from a frustrating puzzle into a straightforward procedure.
Keep your corrected practice problems. Review them before exams. Notice how the same patterns appear repeatedly. Chemistry rewards recognition of these patterns.
Making Chemical Equations Second Nature
Balancing equations stops feeling like work once the process becomes automatic. You’ll reach a point where you see an unbalanced equation and immediately know which coefficient to adjust first.
That fluency comes from repetition, yes, but also from understanding the underlying logic. Chemistry isn’t random. Reactions follow rules. Atoms behave predictably. Your job is to reflect that predictability in properly balanced equations.
The mistakes outlined here trip up thousands of students every semester. You now know what they are and how to avoid them. That knowledge gives you an advantage. Use it. Practice deliberately. Check your work carefully. Build the habit of systematic balancing.
Your next chemistry exam will include equation balancing questions. When you encounter them, remember these principles. Take your time. Follow your process. Trust your training. The equations will balance, your answers will be correct, and your grades will reflect the effort you put into mastering this essential chemistry skill.