Nitrogen makes up 78% of the air you breathe right now, yet most living things can’t use it in that form. This creates one of nature’s most fascinating puzzles: how does this essential element get from the atmosphere into proteins, DNA, and every living cell on Earth? The nitrogen cycle solves this puzzle through a series of chemical transformations that connect air, soil, water, and organisms in ways that sustain all life.
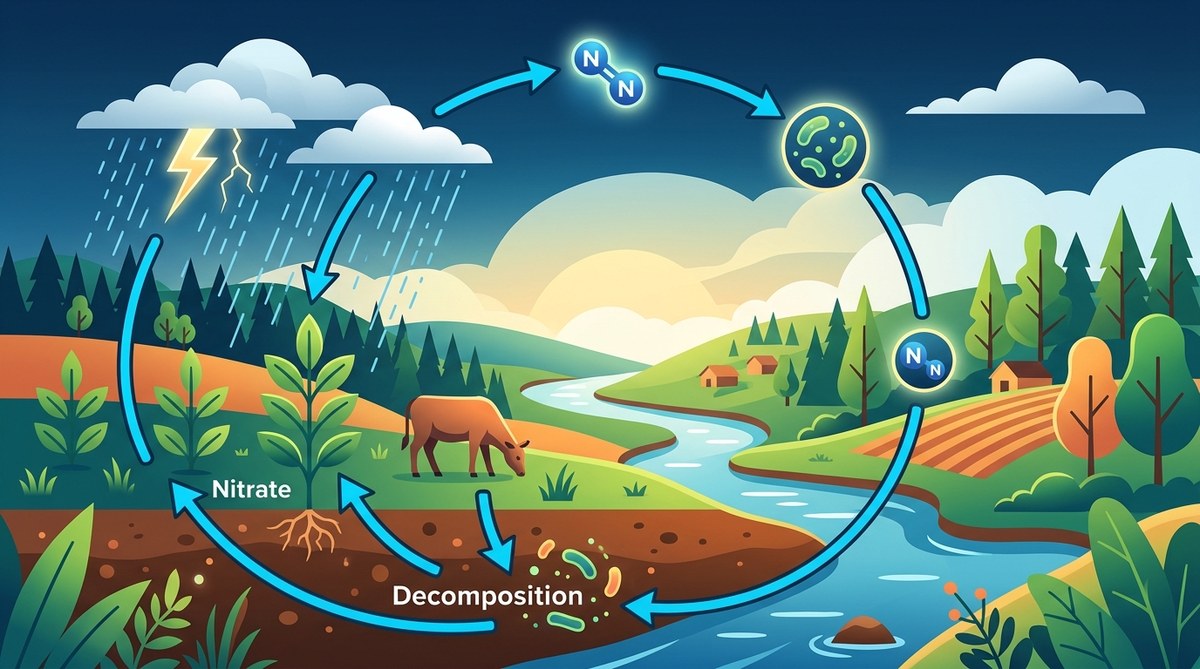
The nitrogen cycle transforms atmospheric nitrogen into usable forms through fixation, moves it through organisms via assimilation, returns it to soil through decomposition, and completes the loop through nitrification and denitrification. This continuous process provides the nitrogen necessary for proteins and DNA while maintaining ecosystem balance through bacterial activity and chemical conversions.
Why Nitrogen Matters for Life
Every protein in your body contains nitrogen. So does every strand of DNA. Plants need nitrogen to grow green leaves and produce seeds. Animals get their nitrogen by eating plants or other animals.
But there’s a problem. Atmospheric nitrogen exists as N₂, two nitrogen atoms bonded so tightly that most organisms can’t break them apart. Only specialized bacteria possess the enzymes needed to crack this bond.
This limitation creates a bottleneck. Life depends on nitrogen, yet 78% of the atmosphere contains a form that life can’t access directly. The nitrogen cycle breaks this bottleneck through five main processes that convert nitrogen between different chemical forms.
The Five Stages of the Nitrogen Cycle
Understanding how nitrogen moves through ecosystems becomes clearer when you break the cycle into distinct stages. Each stage involves specific organisms and chemical reactions.
1. Nitrogen Fixation
Nitrogen fixation converts atmospheric N₂ into ammonia (NH₃). This process requires enormous energy because breaking the triple bond between nitrogen atoms is incredibly difficult.
Certain bacteria perform this task. Rhizobium bacteria live in root nodules of legumes like beans, peas, and clover. Azotobacter bacteria live freely in soil. Cyanobacteria fix nitrogen in aquatic environments.
Lightning also fixes nitrogen. The extreme heat and energy of a lightning strike can break N₂ bonds, allowing nitrogen to combine with oxygen. This creates nitrogen oxides that dissolve in rain and fall to Earth.
Industrial processes fix nitrogen too. The Haber-Bosch process creates ammonia for fertilizers by combining nitrogen and hydrogen under high pressure and temperature. This human intervention now fixes more nitrogen than all natural terrestrial processes combined.
2. Nitrification
Nitrification converts ammonia into forms that plants can absorb more easily. This two-step process requires two different groups of bacteria.
First, Nitrosomonas bacteria oxidize ammonia into nitrite (NO₂⁻). Then Nitrobacter bacteria oxidize nitrite into nitrate (NO₃⁻). Both steps release energy that the bacteria use for growth.
Plants prefer nitrate because it dissolves easily in water and moves through soil to reach roots. Some plants can use ammonia directly, but most rely on nitrifying bacteria to complete this conversion.
This process requires oxygen. Waterlogged soils with low oxygen slow nitrification, which is why wetland plants often show nitrogen deficiency.
3. Assimilation
Assimilation occurs when organisms incorporate nitrogen into their tissues. Plants absorb nitrate and ammonia through their roots, then use these compounds to build amino acids, proteins, and nucleic acids.
Animals can’t absorb nitrogen from soil or air. They must eat plants or other animals to get nitrogen compounds. When you eat a salad, you’re assimilating nitrogen that plants pulled from soil, which bacteria converted from atmospheric N₂.
This stage moves nitrogen up the food chain. A caterpillar eating leaves assimilates plant nitrogen. A bird eating the caterpillar assimilates that same nitrogen again. Each organism rebuilds the nitrogen compounds into forms it needs.
4. Ammonification
Ammonification returns nitrogen to soil when organisms die or produce waste. Decomposer bacteria and fungi break down proteins, DNA, and other nitrogen-containing molecules into ammonia.
When a leaf falls and decays, decomposers release its nitrogen as ammonia. Animal waste contains nitrogen compounds that decomposers also convert to ammonia. Even your own waste goes through this process in sewage treatment or septic systems.
This stage completes a smaller loop within the larger cycle. The ammonia released through ammonification can go through nitrification again, making it available for new plant growth.
5. Denitrification
Denitrification converts nitrate back into N₂ gas, returning nitrogen to the atmosphere. This closes the cycle.
Denitrifying bacteria like Pseudomonas and Clostridium perform this process in oxygen-poor environments. They use nitrate instead of oxygen for respiration, releasing N₂ as a byproduct.
Waterlogged soils, deep ocean sediments, and wetlands provide ideal conditions for denitrification. These bacteria thrive where oxygen is scarce but nitrate is available.
This process might seem wasteful since it removes nitrogen that plants could use. But denitrification prevents nitrogen from accumulating to toxic levels and maintains the balance between atmospheric and terrestrial nitrogen.
How Different Ecosystems Handle Nitrogen
The nitrogen cycle operates everywhere, but the balance between stages varies by ecosystem type.
Forests typically have tight nitrogen cycles. Trees and soil organisms quickly recycle nitrogen from fallen leaves and dead wood. Little nitrogen escapes these systems through runoff or denitrification.
Grasslands experience seasonal nitrogen pulses. Prairie grasses die back in fall, releasing nitrogen through decomposition. Spring rains move this nitrogen into soil where new growth can access it.
Wetlands lose significant nitrogen through denitrification. The waterlogged, oxygen-poor conditions favor denitrifying bacteria. This makes wetlands effective at removing excess nitrogen from water, which is why constructed wetlands help treat agricultural runoff.
Oceans have vast nitrogen reservoirs in deep water. Upwelling brings this nitrogen to surface waters where phytoplankton use it. When these organisms die, their nitrogen sinks back to the depths.
Agricultural fields receive massive nitrogen inputs through fertilizer. Crops remove some nitrogen at harvest, but significant amounts escape through runoff and denitrification. Managing this nitrogen flow challenges farmers worldwide.
A Comparison of Nitrogen Cycle Processes
| Process | Starting Form | Ending Form | Key Organisms | Oxygen Needed? |
|---|---|---|---|---|
| Fixation | N₂ gas | Ammonia (NH₃) | Rhizobium, Azotobacter, cyanobacteria | No |
| Nitrification | Ammonia (NH₃) | Nitrate (NO₃⁻) | Nitrosomonas, Nitrobacter | Yes |
| Assimilation | Nitrate, ammonia | Organic compounds | Plants, animals, microbes | Varies |
| Ammonification | Organic compounds | Ammonia (NH₃) | Decomposer bacteria, fungi | Varies |
| Denitrification | Nitrate (NO₃⁻) | N₂ gas | Pseudomonas, Clostridium | No (uses nitrate instead) |
Human Impact on Nitrogen Cycling
Humans have dramatically altered the nitrogen cycle over the past century. The changes affect every ecosystem on Earth.
Fertilizer production fixes about 120 million metric tons of nitrogen annually. This exceeds all natural terrestrial fixation. Farmers apply this nitrogen to boost crop yields, but plants typically absorb only 30 to 50% of applied fertilizer.
The remaining nitrogen escapes into the environment. Some leaches into groundwater, contaminating drinking water supplies. High nitrate levels in water can cause health problems, particularly for infants. Some nitrogen runs off into streams and rivers, eventually reaching oceans.
Fossil fuel combustion releases nitrogen oxides into the atmosphere. These compounds contribute to acid rain and smog. They also deposit onto soil and water, adding to the nitrogen overload.
“The human alteration of the nitrogen cycle represents one of the most significant environmental changes of the modern era. We’ve roughly doubled the amount of reactive nitrogen entering terrestrial ecosystems, with consequences we’re still working to understand fully.”
Consequences of Excess Nitrogen
Too much nitrogen causes several environmental problems. Understanding these issues helps explain why managing the nitrogen cycle matters.
Eutrophication occurs when excess nitrogen enters water bodies. Algae populations explode, creating dense blooms. When these algae die, decomposers consume oxygen while breaking them down. This creates dead zones where fish and other organisms can’t survive.
The Gulf of Mexico dead zone forms each summer from nitrogen washing down the Mississippi River. It can cover an area the size of New Jersey. Similar dead zones appear in hundreds of locations worldwide.
Soil acidification results from excess nitrogen fertilizer. Nitrification produces hydrogen ions as a byproduct, lowering soil pH. Acidic soils release aluminum and other metals that harm plant roots.
Biodiversity loss follows nitrogen pollution. Some plants thrive in high-nitrogen conditions and outcompete other species. Grasslands that once supported dozens of plant species become dominated by a few nitrogen-loving grasses.
Greenhouse gas emissions include nitrous oxide (N₂O), a byproduct of both nitrification and denitrification. This gas has 300 times the warming potential of carbon dioxide and persists in the atmosphere for over a century.
Practical Ways to Support Healthy Nitrogen Cycling
You can apply nitrogen cycle knowledge in everyday situations, whether you’re gardening, studying for exams, or making consumer choices.
For students preparing for biology exams, focus on the bacterial players in each stage. Questions often ask which organisms perform specific conversions. Knowing that Rhizobium fixes nitrogen while Nitrobacter produces nitrate gives you concrete facts to recall under pressure.
Creating a diagram helps visual learners. Draw boxes for each nitrogen form (N₂, NH₃, NO₂⁻, NO₃⁻, organic N) and arrows showing the processes connecting them. Label each arrow with the process name and key bacteria. This visual map makes the cycle easier to remember than lists of facts.
Gardeners can work with the nitrogen cycle rather than against it. Planting legumes adds nitrogen to soil through fixation. Crop rotation that alternates nitrogen-hungry plants like corn with nitrogen-fixing beans maintains soil fertility naturally. Adding compost provides organic nitrogen that decomposers gradually convert to plant-available forms.
Reducing fertilizer runoff protects water quality. Apply fertilizer only when plants actively grow and can absorb it. Avoid fertilizing before heavy rain that will wash nutrients away. Plant buffer strips of deep-rooted vegetation between fields and streams to capture nitrogen before it reaches water.
Supporting sustainable agriculture makes a difference. Organic farming practices that build soil organic matter create healthier nitrogen cycles. Purchasing food from farms that use cover crops and reduced fertilizer inputs encourages these practices.
Common Misconceptions About Nitrogen
Several misunderstandings about nitrogen and its cycle persist even among students who’ve studied the topic. Clearing these up strengthens your understanding.
Misconception: Plants can use atmospheric nitrogen directly.
Reality: Plants lack the enzymes to break the N₂ triple bond. They depend entirely on bacteria to fix nitrogen into usable forms. This is why legumes with nitrogen-fixing bacteria in their roots grow better than other plants in nitrogen-poor soils.
Misconception: All bacteria perform all nitrogen cycle processes.
Reality: Different bacterial groups specialize in specific conversions. The enzymes for nitrogen fixation differ completely from those for nitrification or denitrification. Some bacteria perform multiple steps, but no single species performs all five main processes. This specialization is similar to how photosynthesis actually works through distinct light and dark reactions.
Misconception: More nitrogen always helps plants grow better.
Reality: Excess nitrogen can harm plants. Too much causes weak, floppy growth vulnerable to pests and disease. It can also create salt stress in soil and prevent plants from absorbing other essential nutrients. Balance matters more than quantity.
Misconception: The nitrogen cycle only occurs in soil.
Reality: Nitrogen cycles through all environments: atmosphere, soil, water, and living organisms. Aquatic nitrogen cycles in oceans, lakes, and rivers mirror terrestrial cycles but involve different organisms and conditions.
Study Tips for Mastering the Nitrogen Cycle
Students often struggle to memorize nitrogen cycle details for exams. These strategies make the information stick better than simple rereading.
Use memory devices for bacterial names. “Rhizo” sounds like “roots” (where Rhizobium lives). “Nitro” appears in Nitrosomonas and Nitrobacter (both involved in nitrification). “Pseudo” means false in Pseudomonas (it falsely uses nitrate as if it were oxygen).
Create comparison tables like the one earlier in this article. Writing out the information yourself, rather than just reading provided tables, strengthens memory formation. Include columns for what you find hardest to remember.
Teach someone else. Explaining the nitrogen cycle to a friend or family member forces you to organize the information clearly. When you stumble explaining a concept, you’ve identified what needs more review.
Connect to real examples. When you see a farm field, think about fixation in soil bacteria. When you notice algae in a pond, consider whether excess nitrogen caused eutrophication. These real-world connections make abstract concepts concrete.
Practice drawing the cycle from memory. Start with a blank page and sketch the cycle without looking at notes. Check your work and try again. Repeat until you can draw and label the entire cycle accurately.
Focus on the “why” behind each step. Understanding that nitrification occurs because bacteria gain energy from oxidizing ammonia makes the process more memorable than simply knowing it happens. The logic behind each step helps you reconstruct the cycle even if you forget specific details.
Nitrogen Cycle Connections to Other Biogeochemical Cycles
The nitrogen cycle doesn’t operate in isolation. It interacts with carbon, water, and phosphorus cycles in ways that affect ecosystem function.
Carbon and nitrogen cycles link closely in soil. Decomposers need both elements. Organic matter with high carbon but low nitrogen decomposes slowly because decomposers can’t get enough nitrogen to build their proteins. Adding nitrogen speeds decomposition, releasing both nitrogen and carbon.
The carbon to nitrogen ratio (C:N ratio) of organic matter determines decomposition rate. Fresh grass clippings with a C:N ratio around 20:1 decompose rapidly. Sawdust with a C:N ratio of 500:1 barely decomposes at all. Composters balance these materials to maintain ideal ratios.
Water movement carries dissolved nitrogen through ecosystems. Rain and irrigation move nitrate down through soil. Runoff carries nitrogen from land to water. Groundwater flow transports nitrogen underground. The water cycle serves as nitrogen’s transportation system.
Phosphorus and nitrogen often limit plant growth together. Adding one without the other may not increase growth if the second remains scarce. This is why fertilizers typically contain both elements. The relationship between these nutrients demonstrates that understanding one cycle requires knowing how it connects to others.
Nitrogen Fixation in Different Environments
While we’ve covered the basics of nitrogen fixation, the process varies significantly across different settings. These variations matter for both natural ecosystems and human agriculture.
Symbiotic fixation in legume root nodules represents the most efficient natural process. The plant provides carbohydrates to fuel the bacteria. The bacteria provide ammonia to the plant. Both benefit from this partnership.
Farmers have exploited this relationship for millennia. Rotating corn with soybeans allows the soybeans to fix nitrogen that the next corn crop uses. This reduces fertilizer needs and maintains soil health.
Free-living fixation by soil bacteria contributes less nitrogen per acre than symbiotic fixation. These bacteria must find their own energy sources, limiting how much nitrogen they can fix. Still, they play important roles in ecosystems without legumes.
Aquatic fixation by cyanobacteria sustains ocean productivity. These photosynthetic bacteria use sunlight for energy while fixing nitrogen. Some form symbiotic relationships with ocean plants, similar to Rhizobium in legume roots.
Rice paddies host cyanobacteria that fix significant nitrogen. Traditional rice farming relied on this natural fertilizer. Modern intensive rice production supplements with industrial fertilizer but still benefits from cyanobacterial fixation.
Industrial fixation through the Haber-Bosch process transformed agriculture. This allows farmers to grow crops on land that couldn’t otherwise support intensive production. However, the process requires substantial energy, mostly from fossil fuels, and contributes to climate change.
Key Nitrogen Compounds and Their Roles
Different nitrogen compounds play distinct roles in the cycle. Recognizing these forms helps you track nitrogen’s path through ecosystems.
- N₂ (nitrogen gas): The atmospheric reservoir, chemically stable and largely unavailable to life
- NH₃ (ammonia): The product of fixation, toxic in high concentrations but usable by some plants
- NH₄⁺ (ammonium): The ionized form of ammonia, binds to soil particles and resists leaching
- NO₂⁻ (nitrite): An intermediate in nitrification, toxic to most organisms in high amounts
- NO₃⁻ (nitrate): Highly mobile in soil, the preferred nitrogen form for most plants
- N₂O (nitrous oxide): A greenhouse gas produced during nitrification and denitrification
- Organic nitrogen: Nitrogen incorporated into proteins, DNA, and other biological molecules
Each compound has different properties affecting how it moves through ecosystems. Nitrate dissolves easily and leaches through soil. Ammonium binds to clay particles and stays put. This is why farmers often use ammonium-based fertilizers in sandy soils where nitrate would wash away.
Why Some Ecosystems Become Nitrogen Limited
Despite nitrogen’s abundance in the atmosphere, many ecosystems experience nitrogen limitation. Several factors create these shortages.
Lack of nitrogen-fixing organisms limits some environments. Desert soils often contain few bacteria capable of fixation. Without these organisms, the nitrogen cycle can’t begin effectively.
Rapid plant growth can deplete available nitrogen faster than the cycle replenishes it. Fast-growing forests in tropical regions often become nitrogen limited because trees incorporate nitrogen into wood that decomposes slowly.
Leaching losses remove nitrogen from some ecosystems faster than inputs can replace it. Heavy rainfall in tropical regions dissolves nitrate and carries it away. Sandy soils that can’t hold nutrients experience similar losses.
Fire volatilizes nitrogen, converting it to gases that escape to the atmosphere. Ecosystems that burn frequently lose nitrogen this way. Some fire-adapted ecosystems have evolved to function with lower nitrogen availability.
Harvesting removes nitrogen from agricultural systems. Each truckload of corn or wheat leaving a farm contains nitrogen that must be replaced. Without inputs through fertilizer or nitrogen fixation, soils gradually become depleted.
Understanding limitation patterns helps explain why different ecosystems have different productivity levels. It also explains why fertilizer application dramatically increases crop yields in many regions.
Bringing the Cycle Together
The nitrogen cycle connects atmosphere, soil, water, and life through a series of transformations that each depend on specific organisms and conditions. Nitrogen fixation converts atmospheric N₂ into ammonia through bacterial action or lightning. Nitrification oxidizes ammonia into nitrate through two bacterial steps. Plants and animals assimilate these compounds into proteins and DNA. Decomposition returns nitrogen to soil as ammonia when organisms die. Denitrification closes the loop by converting nitrate back to N₂ gas.
Each stage matters. Break any link and the cycle falters. Too much human interference and the cycle becomes unbalanced, causing environmental damage. Understanding these connections helps you see ecosystems as integrated systems rather than collections of separate parts. Whether you’re studying for an exam, planning a garden, or simply trying to understand how nature works, the nitrogen cycle provides a perfect example of how elements flow through the living world in patterns that sustain all life.