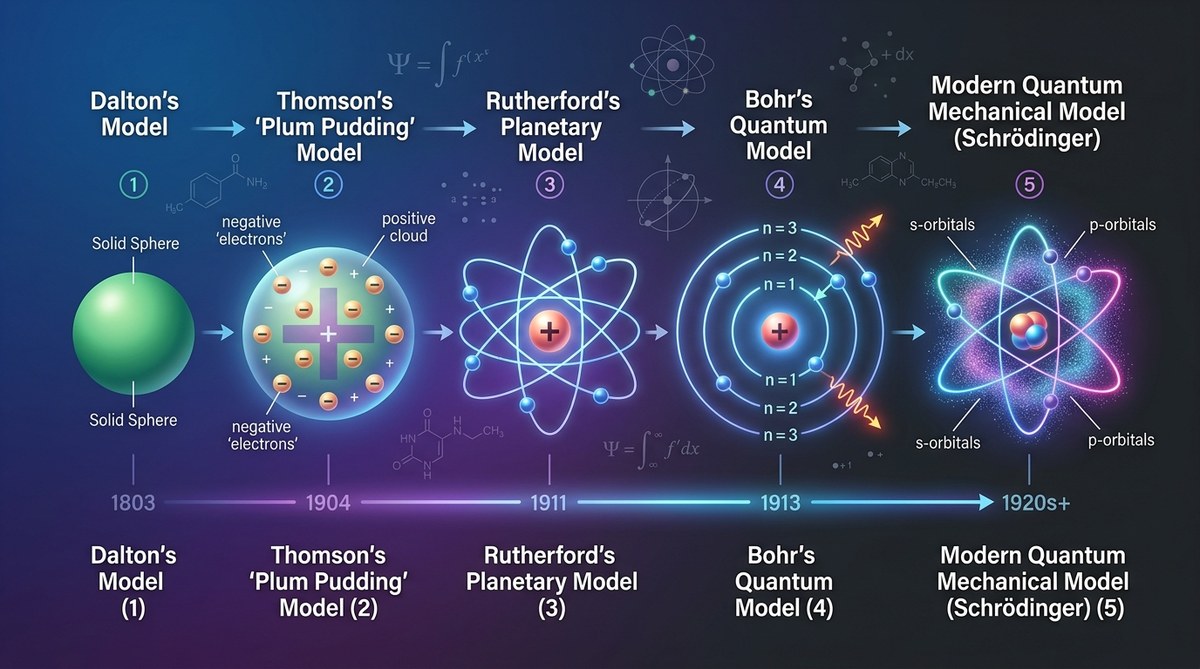
Atoms are the building blocks of matter. Our understanding of what atoms are and how they work has changed dramatically over centuries. From ancient philosophy to cutting-edge quantum physics, the journey of atomic models reflects human curiosity and scientific progress. This story shows how scientists have built on each other’s discoveries, refining the picture of the atom with each breakthrough. Let’s take a close look at how our ideas about atoms have developed over time.
Understanding the evolution of atomic models reveals how scientific theories are refined through experiments and new evidence, leading to a more accurate picture of the atom. From Dalton to quantum mechanics, each model reflects the technological and conceptual advances that shape modern science.
Early Ideas and Philosophical Foundations
Long before scientists could see atoms, ancient philosophers like Democritus proposed that everything is made of tiny, indivisible particles called atomos. These ideas were philosophical and lacked experimental support but laid the groundwork for future scientific inquiry. For centuries, these early notions remained speculative, with no way to test or verify them.
During the 17th and 18th centuries, the Scientific Revolution brought a more systematic approach to understanding matter. Chemists began experimenting with chemical reactions, noticing that elements combine in fixed ratios. These observations hinted at the existence of discrete particles, but the true nature of atoms was still unknown.
Dalton’s Atomic Theory: The First Scientific Model
In 1803, John Dalton revolutionized atomic understanding with his atomic theory. He proposed that each element consists of indivisible atoms, and these atoms have specific weights. Dalton’s model explained chemical reactions as the rearrangement of atoms, which was a major step forward.
Dalton’s atomic model depicted atoms as solid, indivisible spheres, like tiny billiard balls. This simplified picture fit the experimental data available at the time and was widely accepted. However, it could not explain phenomena like electrical conductivity or atomic structure beyond mass ratios.
The Discovery of the Electron and the Plum Pudding Model
In 1897, J.J. Thomson discovered the electron, a tiny charged particle within the atom. This was a huge surprise because Dalton believed atoms were indivisible. Thomson proposed the plum pudding model, where electrons were embedded in a positively charged sphere. This explained how atoms could be divisible and set the stage for more detailed models.
The plum pudding model was useful but had limitations. It couldn’t explain how electrons were arranged or why atoms emitted specific spectra.
Rutherford’s Nuclear Model: The Atom Has a Nucleus
In 1911, Ernest Rutherford performed experiments with alpha particles bombarding a thin gold foil. Most particles went straight through, but some deflected at large angles. Rutherford concluded that atoms have a tiny, dense nucleus containing most of the mass and positive charge.
He proposed a new atomic model with a central nucleus and electrons orbiting around it. This was a big shift from the diffuse plum pudding. It explained atomic stability and led to the idea of a nucleus, but it still couldn’t explain why electrons stayed in orbit or their specific energy levels.
Bohr’s Quantum Model: Electrons in Fixed Orbits
In 1913, Niels Bohr refined Rutherford’s model. He proposed that electrons orbit the nucleus in specific, quantized energy levels. Electrons could jump between levels by absorbing or emitting energy, which explained the discrete spectral lines of hydrogen.
Bohr’s model introduced quantum ideas into atomic theory. Though it successfully explained the hydrogen spectrum, it struggled with more complex atoms and did not account for electron behavior fully.
The Quantum Mechanical Model: Wave-Particle Duality
In the 1920s, scientists like Schrödinger, Heisenberg, and Dirac developed quantum mechanics. Schrödinger’s wave equation described electrons as wave functions, not particles in fixed orbits. Instead of precise paths, electrons are described as regions of probability called orbitals.
This modern atomic model replaces the idea of electrons as tiny planets with a cloud of probability. It accounts for complex atoms and molecules and forms the basis of our current understanding of atomic structure.
How Our Models Evolved Through Science and Technology
The journey from Dalton’s solid spheres to quantum wave mechanics highlights how technological advances have driven scientific understanding. Early microscopes and spectroscopes allowed scientists to observe phenomena that challenged previous models. Today’s tools like particle accelerators and quantum computers continue to refine our picture of the atom.
Scientists constantly test and revise models based on new data. For example, discoveries like the neutron in 1932 and the quark model in the 1960s have added layers of complexity. Each new model incorporates this evidence, making our understanding more precise.
Practical Steps to Understand the Evolution of Atomic Models
- Study historical experiments: Focus on key experiments like Rutherford’s gold foil or the hydrogen spectrum. These experiments provided clues that led to new models.
- Compare models side by side: Use diagrams to visualize how each atomic model looks and functions.
- Understand the scientific method: Recognize how hypotheses are tested, rejected, or refined based on experimental evidence.
Common Techniques and Mistakes in Learning Atomic Models
| Technique | Mistake to Avoid |
|---|---|
| Memorizing models without understanding | Relying solely on memorization instead of grasping concepts |
| Ignoring experimental evidence | Overlooking how experiments shape models |
| Neglecting quantum mechanics | Assuming classical physics explains all atomic behavior |
Key Concepts in Atomic Model Development
- The atom as indivisible (Dalton)
- The nucleus and electrons (Rutherford)
- Quantized energy levels (Bohr)
- Electron clouds and probability (Quantum mechanics)
Expert Advice
“Understanding the evolution of atomic models helps students see science as a dynamic process. Each model is a stepping stone, built upon by new discoveries. Focus on how experimental evidence drives theory development, and you’ll grasp the true nature of scientific progress.”
Modern Atomic Theory and Beyond
Today, atomic models continue to evolve. Quantum mechanics and nuclear physics reveal even smaller particles like quarks and gluons. Researchers are probing the atom’s deepest layers with particle accelerators, pushing the boundary of knowledge.
While the models have become more complex, they also become more accurate. The current picture of the atom includes a nucleus of protons and neutrons surrounded by an electron cloud described by wave functions.
In practical terms, understanding atomic models helps in fields ranging from chemistry and materials science to medicine and energy. Technologies like MRI machines, semiconductors, and nuclear reactors depend on atomic principles.
Final Thoughts: Building Your Atomic Knowledge
Grasping how atomic models have evolved offers insight into the scientific process. Each step reflects a combination of curiosity, experimentation, and theory refinement. As students or enthusiasts, recognizing this progression deepens your appreciation of science and prepares you for advanced topics.
Keep exploring, questioning, and connecting ideas. The journey of understanding atoms is ongoing, and your curiosity fuels future discoveries.
Keep unraveling the atom’s secrets in your scientific journey
Applying this understanding in your studies will help you see science as a continual quest for knowledge. Use diagrams, experiments, and critical thinking to deepen your grasp. Remember, every new discovery builds on the last, and your curiosity can contribute to the next big breakthrough.